分子别名(Synonym)
IL-15,Interleukin-15,MGC9721
表达区间及表达系统(Source)
Human IL-15, premium grade (IL5-H4117) is expressed from E. coli cells. It contains AA Asn 49 - Ser 162 (Accession # P40933-1).
Predicted N-terminus: Met
Human IL-15, premium grade (IL5-H4117), designed for preclinical stage, has the same activity and performance with GMP Human IL-15 (GMP-L15H13), which enables a seamless transition from preclinical development to clinical phases. Premium Grade product offer a cost efficient alternative of GMP Grade products for the early development phase when safety of raw materials is not top priority. By using Premium Grade products in early development phase, you can transition easily into clinical and commercial phase without need to revalidate the raw materials and modify manufacturing process.
蛋白结构(Molecular Characterization)

This protein carries no "tag".
The protein has a calculated MW of 12.9 kDa. The protein migrates as 13 kDa±3 kDa under reducing (R) condition (SDS-PAGE).
内毒素(Endotoxin)
Less than 0.01 EU per μg by the LAL method.
宿主蛋白残留(Host Cell Protein)
<0.5 ng/µg of protein tested by ELISA.
宿主核酸残留(Host Cell DNA)
<0.02 ng/μg of protein tested by DNA Fluorescent Staining method.
无菌(Sterility)
The sterility testing was performed by membrane filtration method.
支原体(Mycoplasma)
Negative.
纯度(Purity)
>95% as determined by SDS-PAGE.
制剂(Formulation)
Lyophilized from 0.22 μm filtered solution in 25 mM Histidine, pH6.2 with trehalose as protectant.
Contact us for customized product form or formulation.
重构方法(Reconstitution)
Please see Certificate of Analysis for specific instructions.
For best performance, we strongly recommend you to follow the reconstitution protocol provided in the CoA.
存储(Storage)
For long term storage, the product should be stored at lyophilized state at -20°C or lower.
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
- -20°C to -70°C for 12 months in lyophilized state;
- -70°C for 3 months under sterile conditions after reconstitution.
电泳(SDS-PAGE)
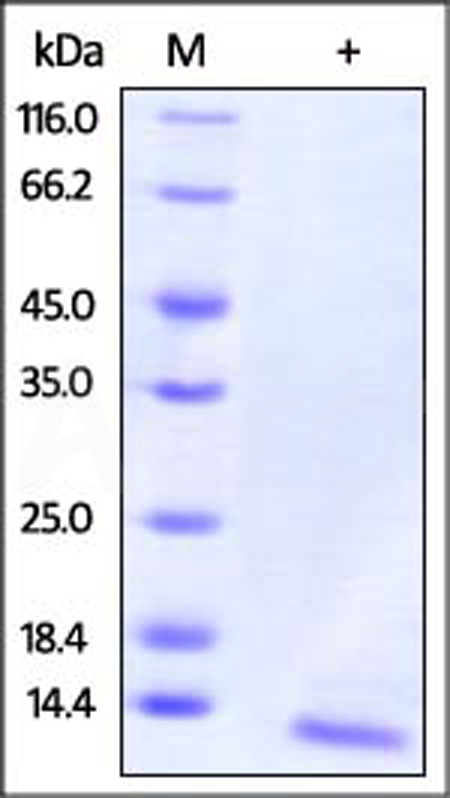
Human IL-15, premium grade on SDS-PAGE under reducing (R) condition. The gel was stained with Coomassie Blue. The purity of the protein is greater than 95%.
活性(Bioactivity)-Bioactivity CELL BASE
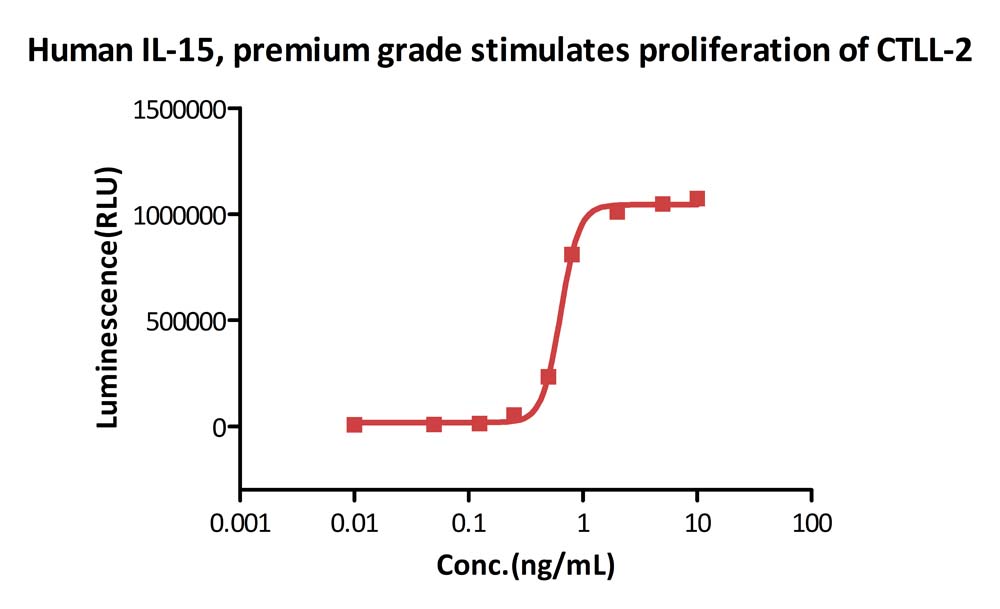
Human IL-15, premium grade (Cat. No. IL5-H4117) stimulates proliferation of CTLL-2 cells. The specific activity of Human IL-15, premium grade is > 8.00ⅹ10^6 IU/mg, which is calibrated against human IL-15 WHO International Standard (NIBSC code: 95/554) (QC tested).
Protocol
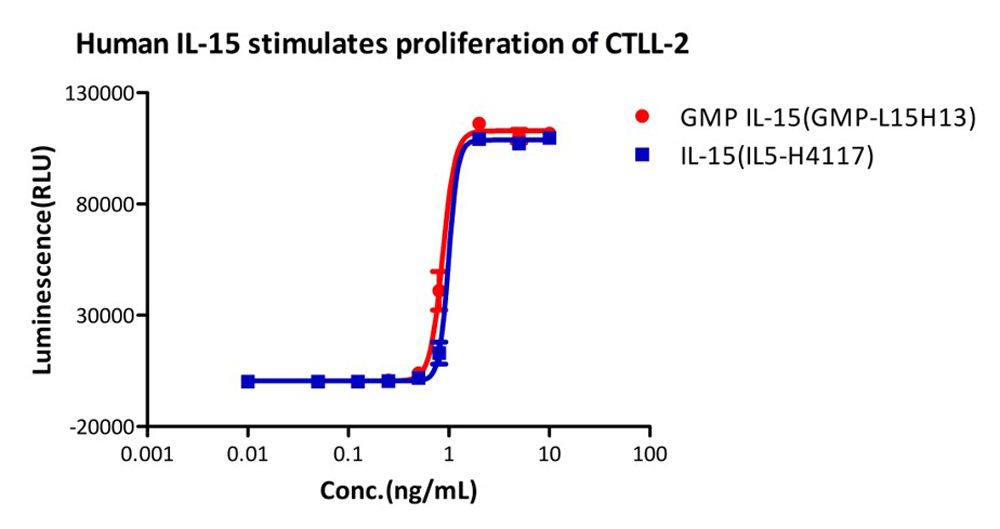
Human IL-15, premium grade (Cat. No. IL5-H4117), designed for preclinical stage, has the same activity and performance with GMP Human IL-15 (Cat. No. GMP-L15H13), which enables a seamless transition from preclinical development to clinical phases.
Protocol
活性(Bioactivity)-ELISA
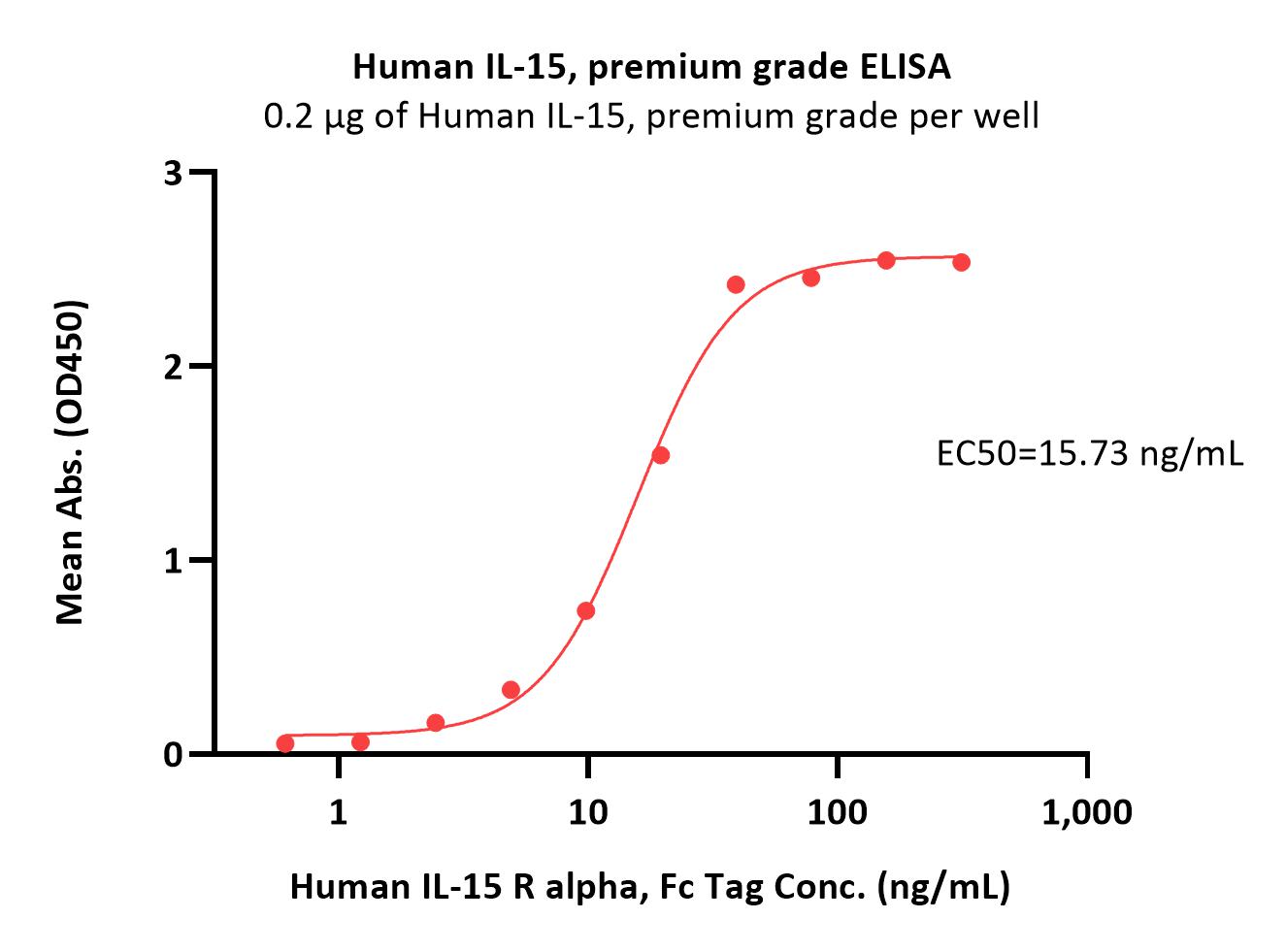
Immobilized Human IL-15, premium grade (Cat. No. IL5-H4117) at 2 μg/mL (100 μL/well) can bind Human IL-15 R alpha, Fc Tag (Cat. No. ILA-H5253) with a linear range of 0.6-39 ng/mL (Routinely tested).
Protocol
活性(Bioactivity)-SPR

Human IL-2 R beta, Fc Tag (Cat. No. ILB-H5253) captured on CM5 chip via anti-human IgG Fc antibody, can bind Human IL-15, premium grade (Cat. No. IL5-H4117) with an affinity constant of 9.5 nM as determined in a SPR assay (Biacore T200) (Routinely tested).
Protocol
活性(Bioactivity)-BLI
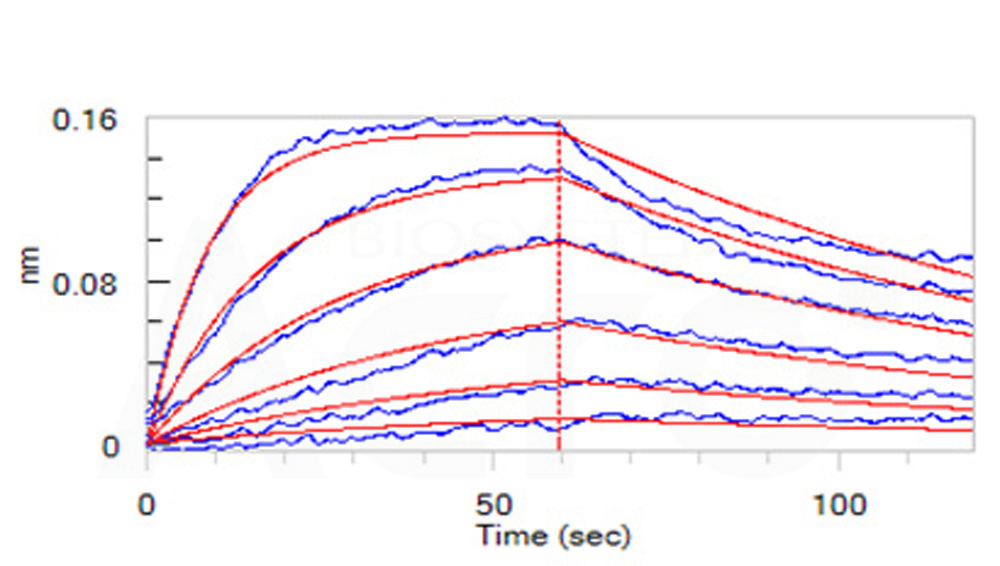
Loaded Human IL-2 R beta, Fc Tag (Cat. No. ILB-H5253) on Protein A Biosensor, can bind Human IL-15, premium grade (Cat. No. IL5-H4117) with an affinity constant of 10.2 nM as determined in BLI assay (ForteBio Octet Red96e) (Routinely tested).
Protocol
背景(Background)
Interleukin 15 is also known as IL15, IL-15, and is a cytokine with structural similarity to IL-2. Like IL-2, IL-15 binds to and signals through the IL-2/IL-15 beta chain (CD122) and the common gamma chain (gamma-C, CD132). IL-15 is secreted by mononuclear phagocytes (and some other cells) following infection by virus(es). This cytokine induces cell proliferation of natural killer cells; cells of the innate immune system whose principal role is to kill virally infected cells. Interleukin 15 (IL-15) regulates T and natural killer (NK) cell activation and proliferation. Survival signals that maintain memory T cells in the absence of antigen are provided by IL-15. This cytokine is also implicated in NK cell development. In rodent lymphocytes, IL-15 prevents apoptosis by inducing an apoptosis inhibitor, BCL2L1/BCL-x(L). IL-15 has been shown to enhance the anti-tumor immunity of CD8+ T cells in pre-clinical models. A phase I clinical trial to evaluate the safety, dosing, and anti-tumor efficacy of IL-15 in patients with metastatic melanoma and renal cell carcinoma (kidney cancer) has begun to enroll patients at the National Institutes of Health.






















































 膜杰作
膜杰作 Star Staining
Star Staining












