描述(Description)
HEK293/Human APP (GFP) Stable Cell Line
应用说明(Application)
Screening β-secretase and γ-secretase inhibitors which stops Aβ peptide formation.
生长特性(Growth Properties)
Adherent
筛选标记(Selection Marker)
Hygromycin B (200 μg/mL)
培养基(Culture Medium)
DMEM medium + 10% FBS
冻存液(Freeze Medium)
10% DMSO + 90% FBS
装量(Quantity)
1 vial contains at least 5×10^6 cells in 1 mL serum-free cryopreservation medium
存储(Storage)
Frozen in liquid nitrogen.
支原体检测(Mycoplasma Testing)
Negative
无菌检测(Sterility Testing)
Negative
使用说明(Instructions for Use)
See data sheet for detailed culturing and assay protocol.
活性(Bioactivity)
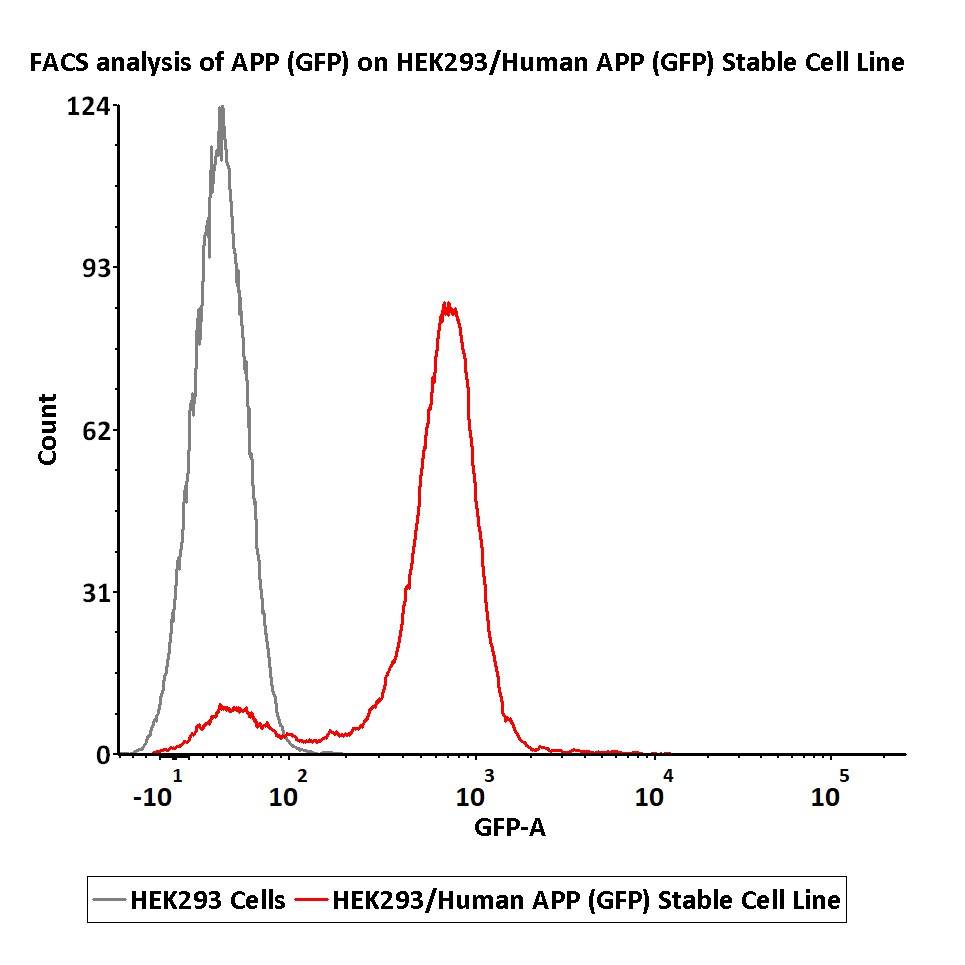
FACS analysis of APP (GFP) on HEK293/Human APP (GFP) Stable Cell Line.
HEK293/Human APP (GFP) Stable Cell Line was red line, negative control HEK293 cells was grey line (QC tested).
Protocol
活性(Bioactivity)-Bioactivity CELL BASE
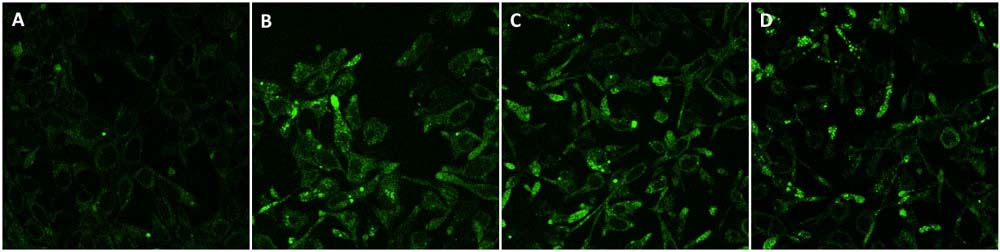
HEK293/Human APP (GFP) Stable Cell Line (Cat. No. CHEK-ATP081) was treated with β-secretase (BACE1) inhibitor and γ-Secretase inhibitor (DAPT) inhibitor, the process of amyloid precursor protein(APP) metabolism by endogenous secretase was inhibited, and the result is shown as fluorescent aggregation of APP-GFP. A. DMSO. B. 10 μm β-secretase (BACE1) inhibitor. C. 10 μM γ -secretase inhibitor (DAPT). D. 5 μm β-secretase (BACE1) inhibitor + 5 μM γ -secretase inhibitor (DAPT) (Routinely tested).
Protocol
如有相关细胞池需求请联系我们
背景(Background)
Amyloid precursor protein (APP) is a type I integral membrane protein ubiquitously expressed in many tissues and concentrated in the synapses of neurons. The majority of APP is cleaved at the plasma membrane by the α-secretase in the non-amyloidogenic pathway. The amyloidogenic pathway starts with β-secretase cleavage by BACE1 on the N-terminal part of the Aβ domain, releasing sAPPβ from a membrane-anchored fragment named βCTF or C103, which is subsequently cleaved by γ-secretase to release Aβ.
License Disclosure
This cell line is provided for research use only. This license does not permit you to share, distribute, sell, sublicense, or otherwise make this cell line available for use to other laboratories, departments, research institutions, hospitals, universities, or biotech companies. The license does not permit modification of this cell line in any way. Inappropriate use or distribution of this cell line will result in revocation of the license. Modifications of this cell line, transfer to another facility, or commercial use of the cell lines may require a separate license and additional fees. AcroBiosystems does not warrant the suitability of this cell line for any particular use, and does not accept any liability in connection with the handling or use of this cell line.






















































 膜杰作
膜杰作 Star Staining
Star Staining











