分子别名(Synonym)
DIF,TNF-alpha,TNFA,TNFSF2,cachexin,cachectin,TNFα
表达区间及表达系统(Source)
Human TNF-alpha, premium grade (TNA-H4211) is expressed from human 293 cells (HEK293). It contains AA Val 77 - Leu 233 (Accession # P01375-1).
Predicted N-terminus: Val 77
It is produced under our rigorous quality control system that incorporates a comprehensive set of tests including sterility and endotoxin tests. Product performance is carefully validated and tested for compatibility for cell culture use or any other applications in the early preclinical stage. When ready to transition into later clinical phases, we also offer a custom GMP protein service that tailors to your needs. We will work with you to customize and develop a GMP-grade product in accordance with your requests that also meets the requirements for raw and ancillary materials use in cell manufacturing of cell-based therapies.
Request for sequence
蛋白结构(Molecular Characterization)

This protein carries no "tag".
The protein has a calculated MW of 17.4 kDa. The protein migrates as 17 kDa under reducing (R) condition (SDS-PAGE).
内毒素(Endotoxin)
Less than 0.02 EU per μg by the LAL method.
无菌(Sterility)
The sterility testing was performed by membrane filtration method.
支原体(Mycoplasma)
Negative.
纯度(Purity)
>95% as determined by SDS-PAGE.
>95% as determined by SEC-MALS.
制剂(Formulation)
Lyophilized from 0.22 μm filtered solution in PBS, pH7.4 with trehalose as protectant.
Contact us for customized product form or formulation.
重构方法(Reconstitution)
Please see Certificate of Analysis for specific instructions.
For best performance, we strongly recommend you to follow the reconstitution protocol provided in the CoA.
存储(Storage)
For long term storage, the product should be stored at lyophilized state at -20°C or lower.
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
- -20°C to -70°C for 24 months in lyophilized state;
- -70°C for 3 months under sterile conditions after reconstitution.
电泳(SDS-PAGE)

Human TNF-alpha, premium grade on SDS-PAGE under reducing (R) condition. The gel was stained with Coomassie Blue. The purity of the protein is greater than 95%.
SEC-MALS
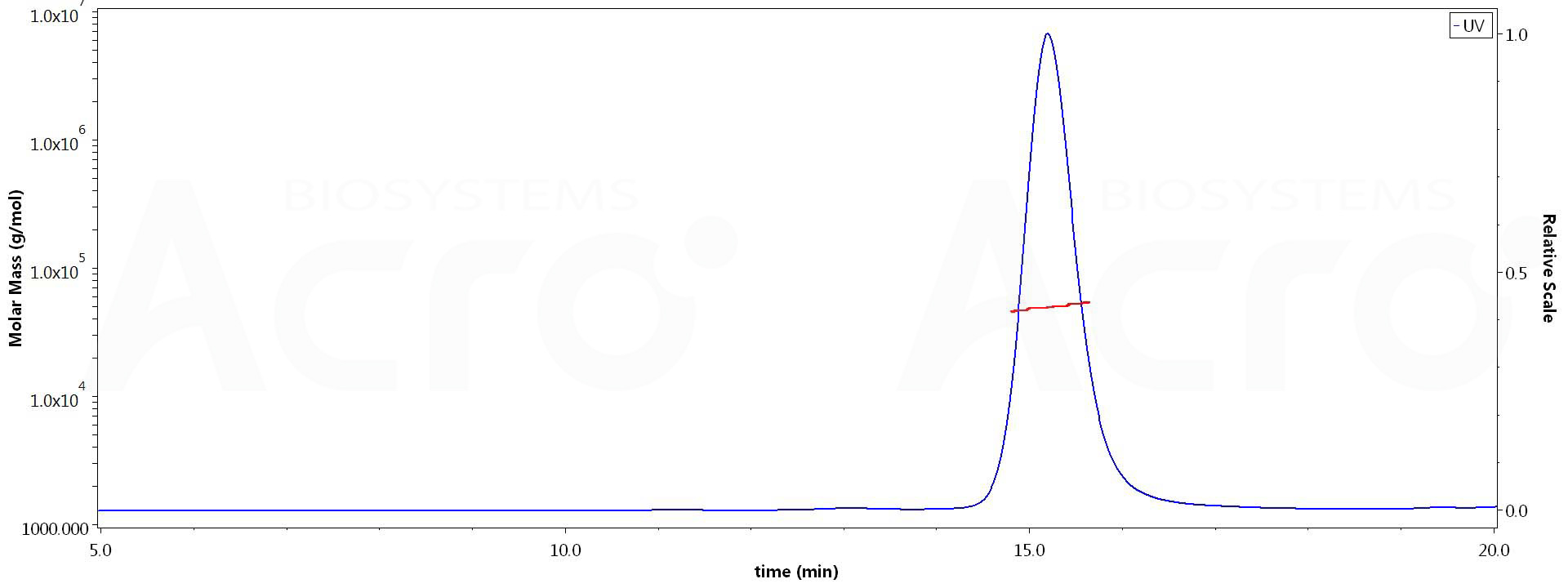
The purity of Human TNF-alpha, premium grade (Cat. No. TNA-H4211) is more than 95% and the molecular weight of this protein is around 47-60 kDa verified by SEC-MALS.
Report
活性(Bioactivity)-Bioactivity CELL BASE
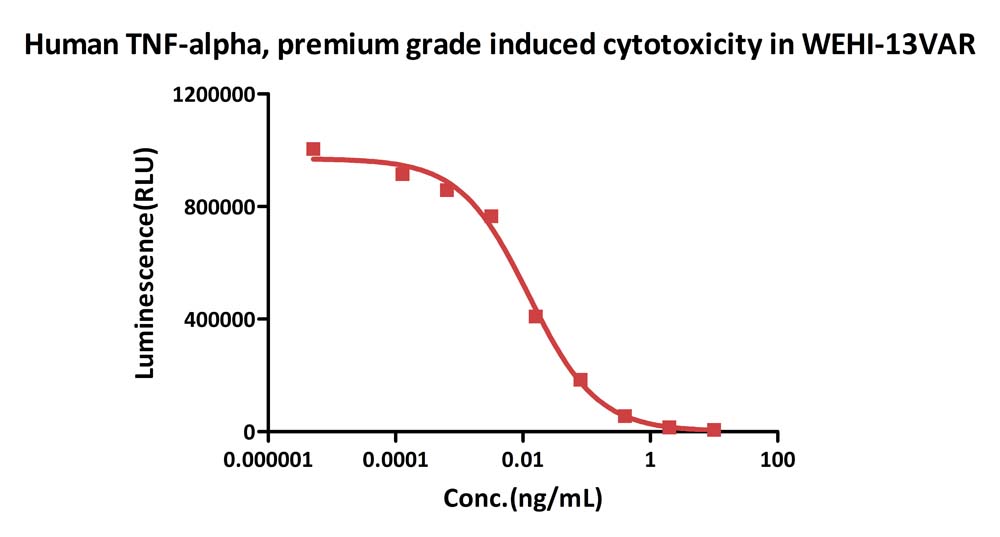
Human TNF-alpha, premium grade (Cat. No. TNA-H4211) induces cytotoxicity effect on the WEHI-13VAR cells in the presence of the metabolic inhibitor actinomycin D. The specific activity of Human TNF-alpha, premium grade is > 2.00×10^7 IU/mg, which is calibrated against human TNF-alpha WHO International Standard (NIBSC code: 12/154) (QC tested).
Protocol
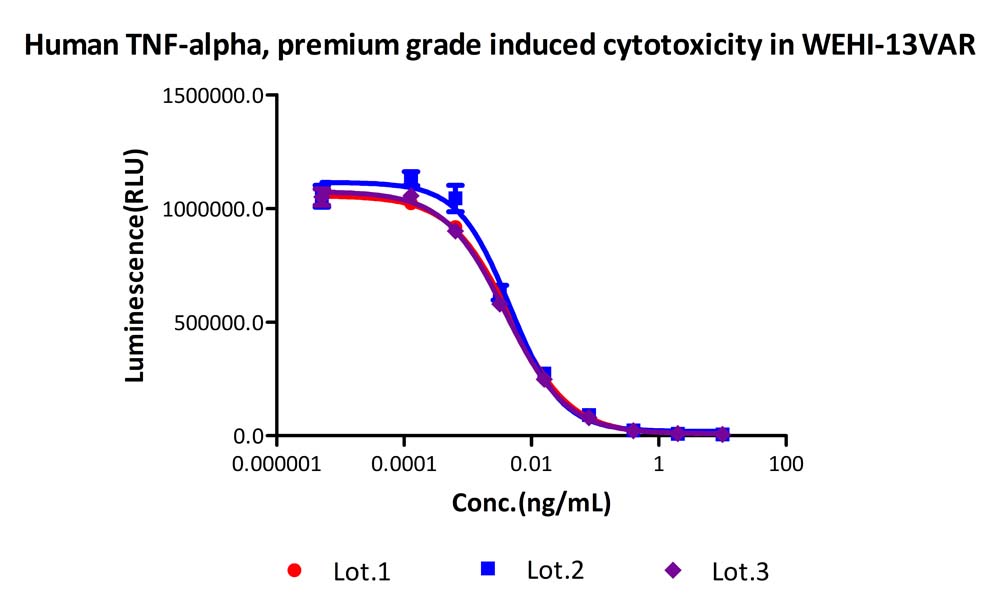
Activity of three different production batches of Human TNF-alpha, premium grade (Cat. No. TNA-H4211).

Neutralization assay shows that the cytotoxicity effect of Human TNF-alpha, premium grade (Cat. No. TNA-H4211) was inhibited by increasing concentration of Adalimumab. The concentration of TNF-alpha used is 1 ng/mL. The IC50 is 29 ng/mL (Routinely tested).
Protocol
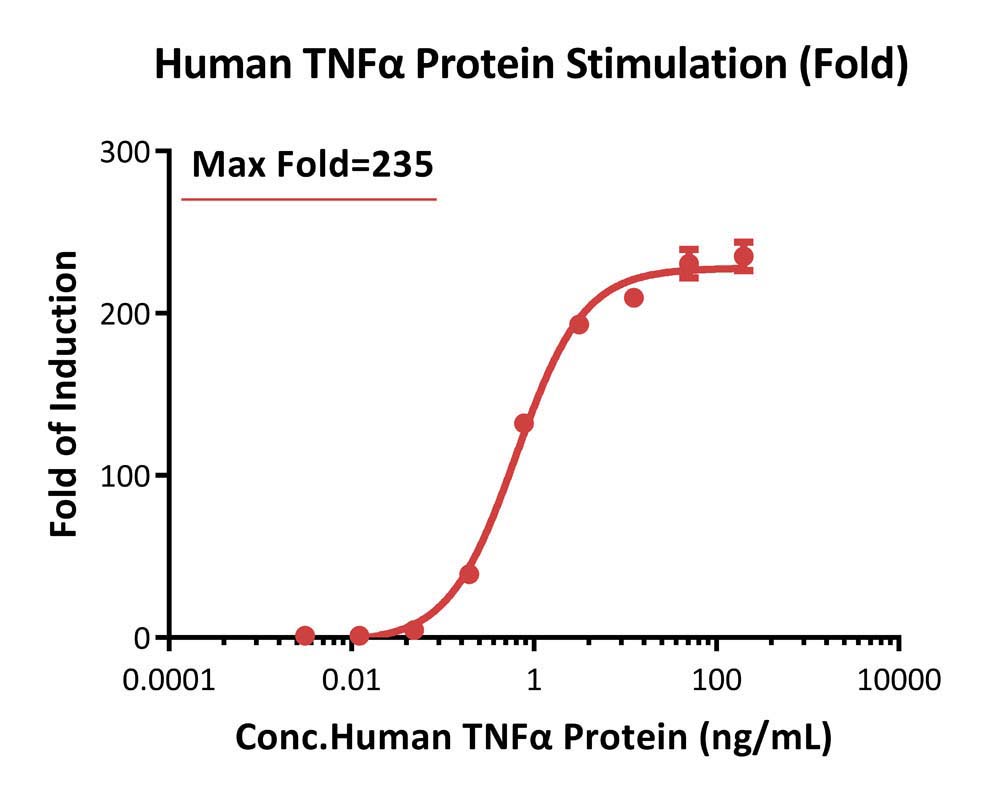
The NF-kB (Luc) HEK293 Reporter Cell was stimulated with serial dilutions of Human TNF-alpha, premium grade (Cat. No. TNA-H4211). The max induction fold was approximately 235 (Routinely tested).
Protocol
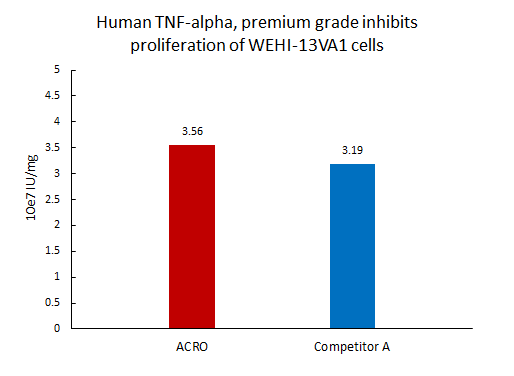
The activity of Human TNF-alpha, premium grade (TNA-H4211) was higher than other competing products.
稳定性(Stability)
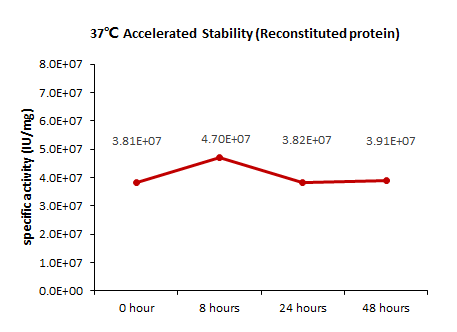
The Cell based assay shows that Human TNF-alpha, premium grade (TNA-H4211) is stable at 37°C for 48 hours.
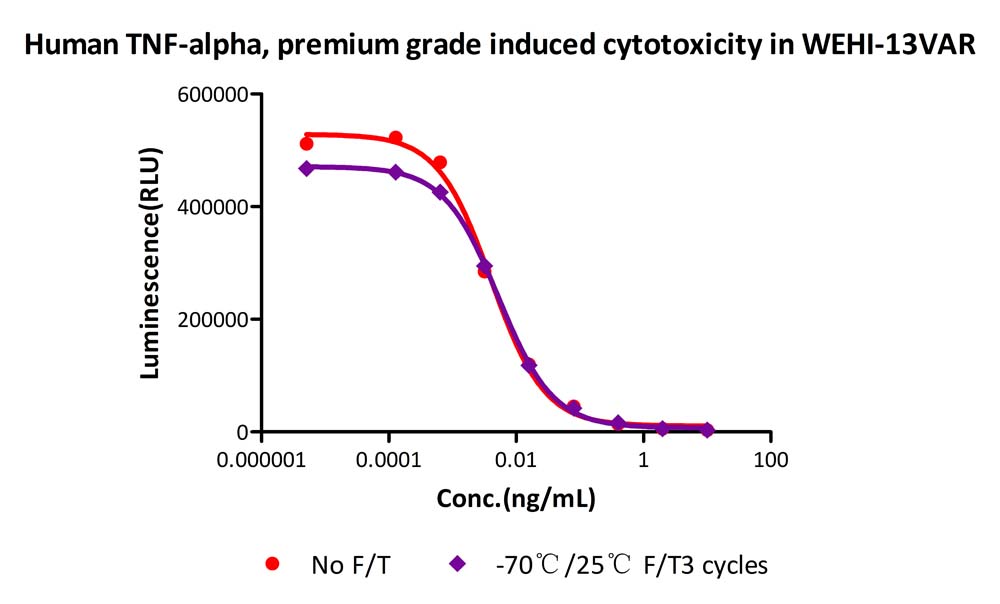
The Cell based assay shows that Human TNF-alpha, premium grade (TNA-H4211) is stable after freezing and thawing 3 times.
活性(Bioactivity)-ELISA
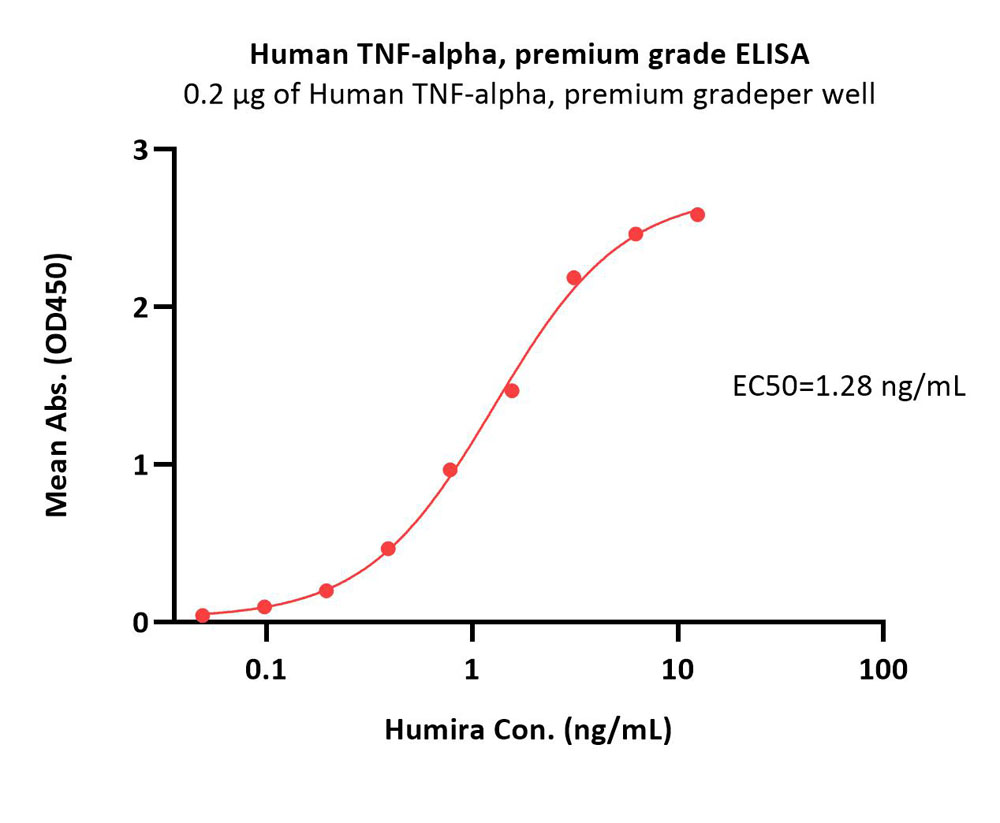
Immobilized Human TNF-alpha, premium grade (Cat. No. TNA-H4211) at 2 μg/mL (100 μL/well) can bind Humira with a linear range of 0.2-2 ng/mL (QC tested).
Protocol
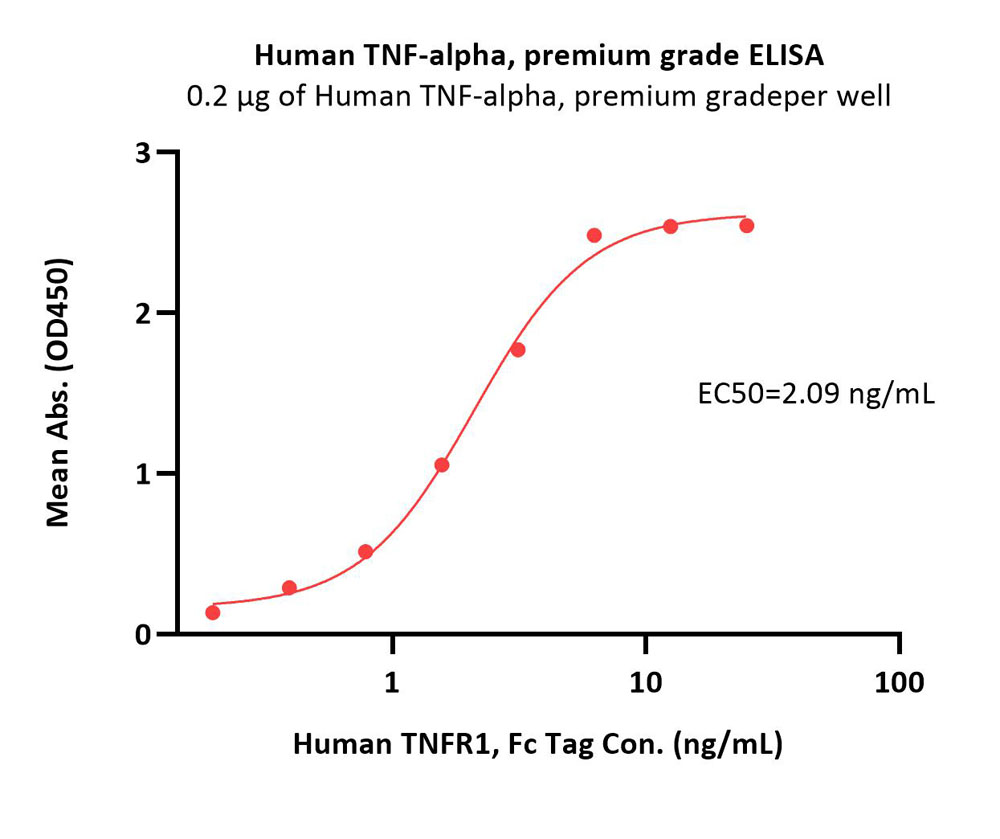
Immobilized Human TNF-alpha, premium grade (Cat. No. TNA-H4211) at 2 μg/mL (100 μL/well) can bind Human TNFR1, Fc Tag (Cat. No. TN1-H5251) with a linear range of 0.2-3 ng/mL (Routinely tested).
Protocol
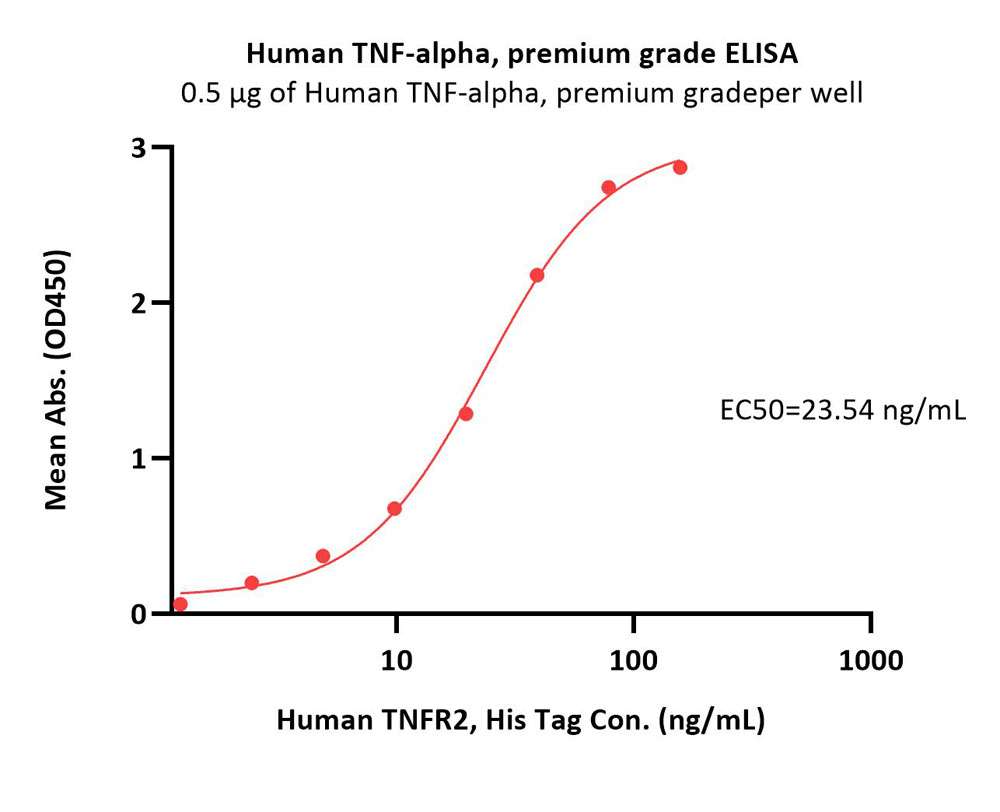
Immobilized Human TNF-alpha, premium grade (Cat. No. TNA-H4211) at 5 μg/mL (100 μL/well) can bind Human TNFR2, His Tag (Cat. No. TN2-H5227) with a linear range of 5-39 ng/mL (Routinely tested).
Protocol
活性(Bioactivity)-SPR
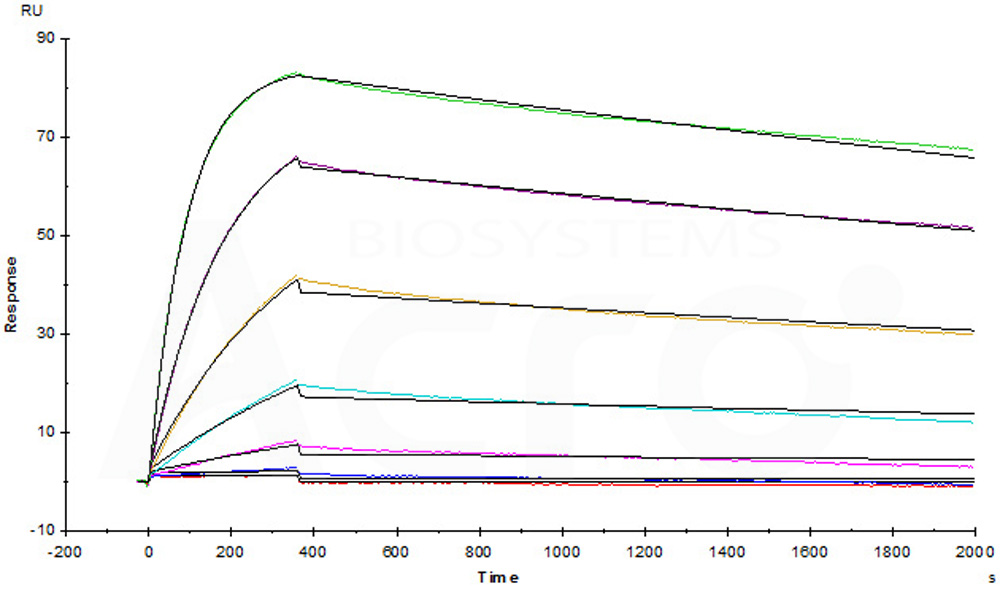
Humira (Adalimumab) captured on CM5 chip via anti-human IgG Fc antibodies surface, can bind Human TNF-alpha, premium grade (Cat. No. TNA-H4211) with an affinity constant of 0.255 nM as determined in a SPR assay (Biacore T200) (Routinely tested).
Protocol
背景(Background)
Tumor necrosis factor alpha (TNFα) is a cytokine produced primarily by monocytes and macrophages. It is found in synovial cells and macrophages in the tissues.The primary role of TNFα is in the regulation of immune cells. TNFα is able to induce apoptotic cell death, to induce inflammation, and to inhibit tumorigenesis and viral replication. Dysregulation of TNFα production has been implicated in a variety of human diseases, including major depression, Alzheimer's disease and cancer. Recombinant TNFα is used as an immunostimulant under the INN tasonermin. TNFα can be produced ectopically in the setting of malignancy and parallels parathyroid hormone both in causing secondary hypercalcemia and in the cancers with which excessive production is associated.






















































 膜杰作
膜杰作 Star Staining
Star Staining












