-
SARS-CoV-2 mRNA-vaccine candidate; COReNAPCIN®, induces robust humoral and cellular immunity in mice and non-human primates
Authors: Alimohammadi R, Porgoo M, Eftekhary M, et al.
Journal: NPJ vaccines 2022
Application: ELISA
Request for Full-text
-
Comparative characterization of antibody responses induced by Ad5-vectored spike proteins of emerging SARS-CoV-2 VOCs
Authors: Wang, Busen et al
Journal: Signal Transduct Target Ther 2022
Application: ELISA
Request for Full-text
-
Preclinical immunogenicity and efficacy of a candidate COVID-19 vaccine based on a vesicular stomatitis virus-SARS-CoV-2 chimera
Authors: Espeseth AS, Yuan M, Citron M, et al.
Journal: EBioMedicine 2022
Application: Binding Assay
Request for Full-text
-
Amphiphile-CpG vaccination induces potent lymph node activation and COVID-19 immunity in mice and non-human primates
Authors: Seenappa LM, Jakubowski A, Steinbuck MP, et al.
Journal: NPJ Vaccines 2022
Application: Binding Assay
Request for Full-text
-
SARS‐CoV‐2 spike protein aggregation is triggered by bacterial lipopolysaccharide
Authors: Petrlova, Jitka et al.
Journal: FEBS Lett 2022
Application: Binding Assay
Request for Full-text
-
Purification of Emu IgY for Therapeutic and Diagnostic Use Based on Monoclonal Secondary Antibodies Specific to Emu IgY
Authors: Kouya Yamaki, Kiyoe Ohta, Norihiro Kobayashi
Journal: Biological and Pharmaceutical Bulletin 2022
Application: Immunization
Request for Full-text
-
An ACE2-IgG4 Fc Fusion Protein Demonstrates Strong Binding to All Tested SARS-CoV-2 Variants and Reduced Lung Inflammation in Animal Models of SARS-CoV-2 and Influenza
Authors: Emmanuel Y. Merigeon, Dong Yang, et al.
Journal: Pathogens and Immunity 2022
Application: Binding analysis. BLI
Request for Full-text
-
Unadjuvanted intranasal spike vaccine elicits protective mucosal immunity against sarbecoviruses
Authors: Tianyang Mao, Benjamin Israelow, Mario A. Peña-Hernández, et al.
Journal: RESEARCH 2022
Application: Vaccination
Request for Full-text
-
Omicron infection increases IgG binding to spike protein of predecessor variants
Authors: Mahalingam G, Periyasami Y, Arjunan P, et al.
Journal: Journal of Medical Virology 2022
Application: ELISA
Request for Full-text
-
Safety and immunogenicity of the bi-cistronic GLS-5310 COVID-19 DNA vaccine delivered with the GeneDerm suction device
Authors: Kim WJ, Roberts CC, Song JY, et al.
Journal: International Journal of Infectious Diseases 2022
Application: ELISA
Request for Full-text
-
A potent synthetic nanobody with broad-spectrum activity neutralizes SARS-CoV-2 virus and the Omicron variant BA. 1 through a unique binding mode
Authors: Zhao D, Liu L, Liu X, et al.
Journal: Journal of Nanobiotechnology 2022
Application: ELISA
Request for Full-text
-
Early CD4+ T cell responses induced by the BNT162b2 SARS-CoV-2 mRNA vaccine predict immunological memory
Authors: Jie Bai, Asako Chiba, Goh Murayama, et al.
Journal: scientific reports 2022
Application: Fow cytometry
Request for Full-text
-
SARS-CoV-2 spike protein induces a differential monocyte activation that may contribute to age bias in COVID-19 severity
Authors: InesAit‑Belkacem1, Celia CartagenaGarcía1, Ewa Millet‑Wallisky, et al.
,
Journal: Scientifc Reports 2022
Application: Cell culture
Request for Full-text
-
Immune response and protective efficacy of the SARS-CoV-2 recombinant spike protein vaccine S-268019-b in mice
Authors: Tomoyuki Homma1,Noriyo Nagata,Masayuki Hashimoto, et al.
Journal: Scientifc Reports 2022
Application: ELISA
Request for Full-text
-
Interim analysis from a phase 2 randomized trial of EuCorVac-19: a recombinant protein SARS-CoV-2 RBD nanoliposome vaccine
Authors: Lovell J F, Baik Y O, Choi S K, et al.
Journal: BMC medicine 2022
Application: ELISA
Request for Full-text
-
Novel super-neutralizing antibody UT28K is capable of protecting against infection from a wide variety of SARS-CoV-2 variants
Authors: T Ozawa, H Tani, Y Anraku, et. al
Journal: mAbs 2022
Application: ISAAC
Request for Full-text
-
mRNA vaccine induces cytotoxic CD8+ T-cell cross-reactivity against SARS-CoV-2 Omicron variant and regulates COVID-19 severity
Authors: T Nogimori, K Suzuki, Y Masuta, et. al
Journal: Research Square 2022
Application: ELISA
Request for Full-text
-
Clinical grade ACE2 as a universal agent to block SARS‐CoV‐2 variants
Authors: V Monteil, B Eaton, E Postnikova, et. al
Journal: EMBO Molecular Medicine 2022
Application: SPR
Request for Full-text
-
Antibody-dependent enhancement (ADE) of SARS-CoV-2 pseudoviral infection requires FcγRIIB and virus-antibody complex with bivalent interaction
Authors: S Wang, J Wang, X Yu et al.
Journal: mBio 2022
Application: SPR
Request for Full-text
-
Follicular T cells optimize the germinal center response to SARS-CoV-2 protein vaccination in mice
Authors: CB Cavazzoni, BL Hanson, MA Podestà et al.
Journal: Cell Reports 2022
Application: ELISA for antibody specificity
Request for Full-text
-
Rapid discovery of diverse neutralizing SARS-CoV-2 antibodies from large-scale synthetic phage libraries
Authors: TZ Yuan, P Garg, L Wang et al.
Journal: mAbs 2022
Application: SPR
Request for Full-text
-
Healthy humans can be a source of antibodies countering COVID-19
Authors: N Velappan, HB Nguyen, S Micheva-Viteva, et al.
Journal: Bioengineered 2022
Application: ELISA
Request for Full-text
-
A lethal mouse model for evaluating vaccine-associated enhanced respiratory disease during SARS-CoV-2 infection
Authors: N Iwata-Yoshikawa, N Shiwa, T Sekizuka, et al.
Journal: Science Advances 2022
Application: ELISA
Request for Full-text
-
Three SARS-CoV-2 antibodies provide broad and synergistic neutralization against variants of concern, including Omicron
Authors: S Wang, H Sun, Y Zhang, et al.
Journal: Cell Reports 2022
Application: Microscopy
Request for Full-text
-
Broad ultra-potent neutralization of SARS-CoV-2 variants by monoclonal antibodies specific to the tip of RBD
Authors: Ma H, Guo Y, Tang H, et al.
Journal: Cell discovery 2022
Application: ELISA
Request for Full-text
-
No evidence of fetal defects or anti-syncytin-1 antibody induction following COVID-19 mRNA vaccination
Authors: Lu-Culligan A, Tabachnikova A, Pérez-Then E, et al.
Journal: PLoS biology 2022
Application: ELISA
Request for Full-text
-
Neutralizing antibodies against the SARS-CoV-2 Delta and Omicron variants following heterologous CoronaVac plus BNT162b2 booster vaccination
Authors: Pérez-Then E, Lucas C, Monteiro VS, et al.
Journal: Nature medicine 2022
Application: ELISA
Request for Full-text
-
Preclinical study of a DNA vaccine targeting SARS-CoV-2
Authors: Hayashi H, Sun J, Yanagida Y, et al.
Journal: Curr Res Transl Med 2022
Application: ELISA
Request for Full-text
-
SARS-CoV-2 vaccination induces immunological T cell memory able to cross-recognize variants from Alpha to Omicron
Authors: Tarke A, Coelho CH, Zhang Z, et al.
Journal: Cell 2022
Application: ELISA
Request for Full-text
-
Evidence for investigating GSK-3 inhibitors as potential therapeutics for severe COVID-19
Authors: Ghazanfari, Davoud et al.
Journal: Biochem Biophys Res Commun 2022
Application: Cell-based assay
Request for Full-text
-
Human-Immune-System (HIS) humanized mouse model (DRAGA: HLA-A2. HLA-DR4. Rag1KO. IL-2RγcKO. NOD) for COVID-19
Authors: TD Brumeanu, P Vir, AF Karim, et al.
Journal: Human Vaccines & Immunotherapeutics 2022
Application: ELISA
Request for Full-text
-
Few SARS-CoV-2 infections detected in Newfoundland and Labrador in the absence of Public Health Laboratory-based confirmation
Authors: DP Ings, KM Hatfield, KE Fifield, et al.
Journal: PLoS ONE 2022
Application: ELISA
Request for Full-text
-
T-cell expression of angiotensin-converting enzyme 2 and binding of severe acute respiratory coronavirus 2
Authors: JL Welch, J Xiang, Q Chang, et al.
Journal: The Journal of Infectious Diseases 2022
Application: MTT assay
Request for Full-text
-
First-in-class trispecific VHH-Fc based antibody with potent prophylactic and therapeutic efficacy against SARS-CoV-2 and variants
Authors: A Titong, S Gallolu, et al.
Journal: Scientific Reports 2022
Application: BLI
Request for Full-text
-
DNA Vaccine Administered by Cationic Lipoplexes or by In Vivo Electroporation Induces Comparable Antibody Responses against SARS-CoV-2 in Mice
Authors: Allegra Peletta, Eakachai Prompetchara, et al.
Journal: Vaccines 2021
Application: ELISA
Request for Full-text
-
Lentil lectin derived from Lens culinaris exhibit broad antiviral activities against SARS-CoV-2 variants
Authors: Wang W, Li Q, Wu J, et al.
Journal: Emerg Microbes Infect 2021
Application: SPR
Request for Full-text
-
Directed evolution of potent neutralizing nanobodies against SARS-CoV-2 using CDR-swapping mutagenesis
Authors: Zupancic JM, Desai AA, Schardt JS, et al.
Journal: Cell chemical biology 2021
Application: Binding Assay
Request for Full-text
-
SARS-CoV-2 induces transcription of human endogenous retrovirus RNA followed by type W envelope protein expression in human lymphoid cells.
Authors: Charvet B, Mazelier M, Brunel J, et al.
Journal: Research Square 2021
Application: Cell-based assay
Request for Full-text
-
Robust SARS-CoV-2 infection in nasal turbinates after treatment with systemic neutralizing antibodies
Authors: Zhou D, Chan JF, Zhou B, et al.
Journal: Cell Host Microbe 2021
Application: SPR
Request for Full-text
-
Nano-size dependence in the adsorption by the SARS-CoV-2 spike protein over gold colloid
Authors: Yokoyama K, Ichiki A.
Journal: Colloids Surf A Physicochem Eng Asp 2021
Application: SPR
Request for Full-text
-
SARS-CoV-2 D614G spike mutation increases entry efficiency with enhanced ACE2-binding affinity
Authors: Ozono S, Zhang Y, Ode H, et al.
Journal: Nature communications 2021
Application: BLI
Request for Full-text
-
Spectroscopic investigation on the affinity of SARS-CoV-2 spike protein to gold nano-particles
Authors: Kazushige Yokoyama, Akane Ichiki
Journal: Colloid and Interface Science Communications 2020
Application: SPR
Request for Full-text
-
HDL-scavenger receptor B type 1 facilitates SARS-CoV-2 entry
Authors: Wei C, Wan L, Yan Q, et al.
Journal: Nature metabolism 2020
Application: SPR & FACS & Confocal microscopy
Request for Full-text
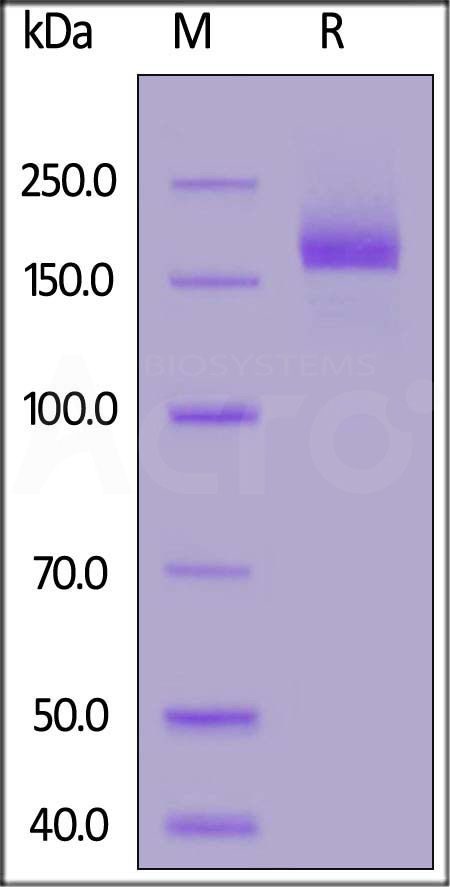
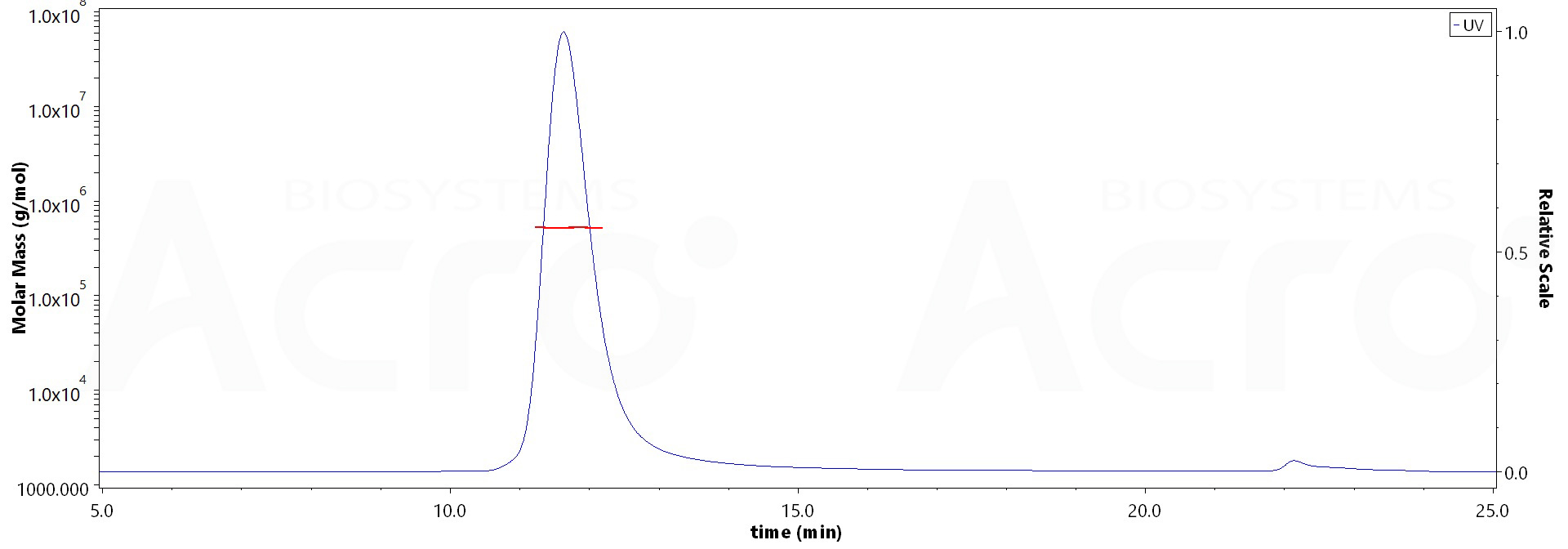

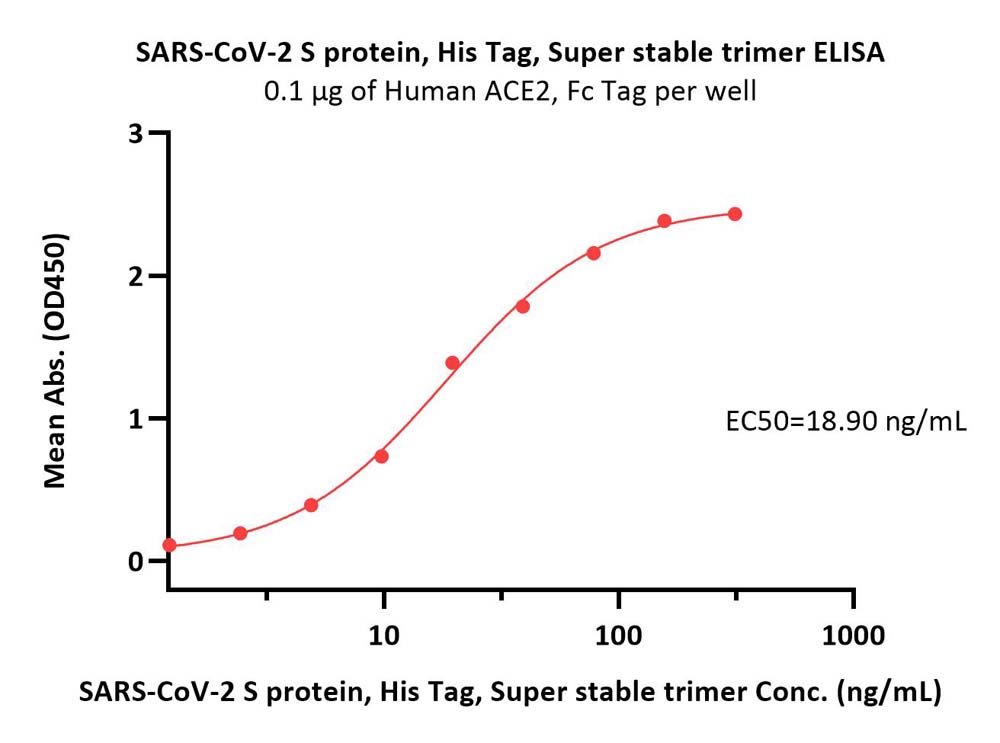
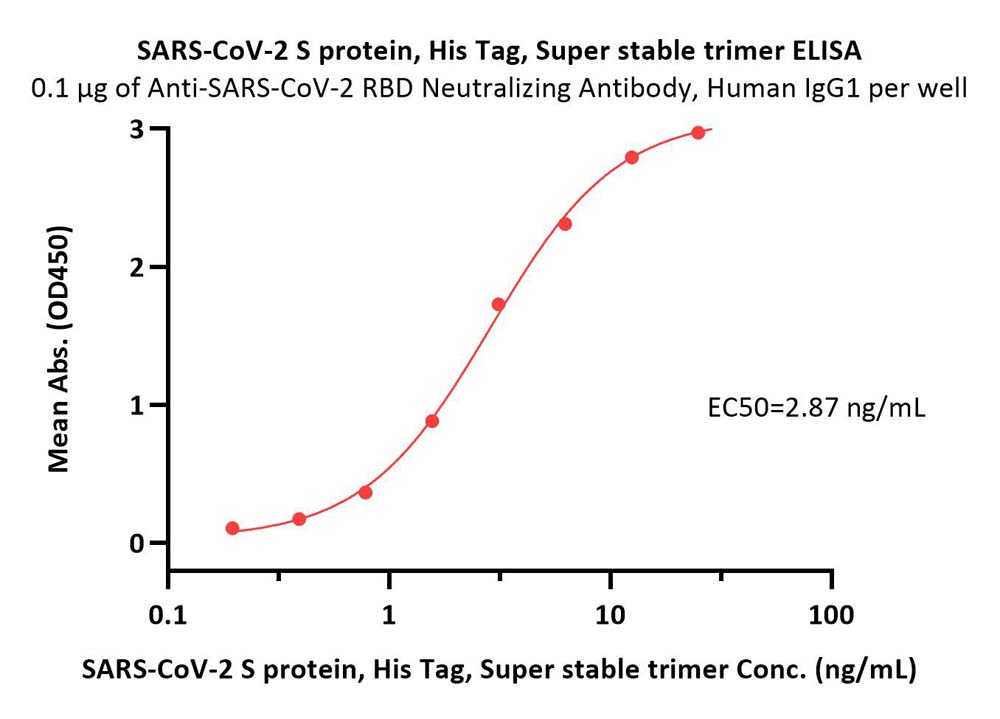






















































 膜杰作
膜杰作 Star Staining
Star Staining












