分子别名(Synonym)
GPC3,OCI5,Glypican-3,GTR2-2,MXR7,DGSX,SDYS ,SGB,SGBS,SGBS1
表达区间及表达系统(Source)
Cynomolgus Glypican 3, His Tag (GP3-C5225) is expressed from human 293 cells (HEK293). It contains AA Gln 25 - His 559 (Accession # XP_005594665.1).
Predicted N-terminus: Gln 25 & Ser 359
Request for sequence
蛋白结构(Molecular Characterization)

This protein carries a polyhistidine tag at the C-terminus.
This protein contains a furin-like convertase cleavage site, 355-RQYR-358, and will be processed into N and C-terminal fragment with calculated MW of 38.1 kDa and 24.6 kDa respectively. The N and C‑terminal fragment as 42 kDa and 66-130 kDa under reducing (R) condition (SDS-PAGE) due to glycosylation.
内毒素(Endotoxin)
Less than 1.0 EU per μg by the LAL method.
纯度(Purity)
>85% as determined by SDS-PAGE.
制剂(Formulation)
Lyophilized from 0.22 μm filtered solution in PBS, pH7.4 with trehalose as protectant.
Contact us for customized product form or formulation.
重构方法(Reconstitution)
Please see Certificate of Analysis for specific instructions.
For best performance, we strongly recommend you to follow the reconstitution protocol provided in the CoA.
存储(Storage)
For long term storage, the product should be stored at lyophilized state at -20°C or lower.
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
- -20°C to -70°C for 12 months in lyophilized state;
- -70°C for 3 months under sterile conditions after reconstitution.
电泳(SDS-PAGE)

Cynomolgus Glypican 3, His Tag on SDS-PAGE under reducing (R) condition. The gel was stained with Coomassie Blue. The purity of the protein is greater than 85%.
活性(Bioactivity)-ELISA
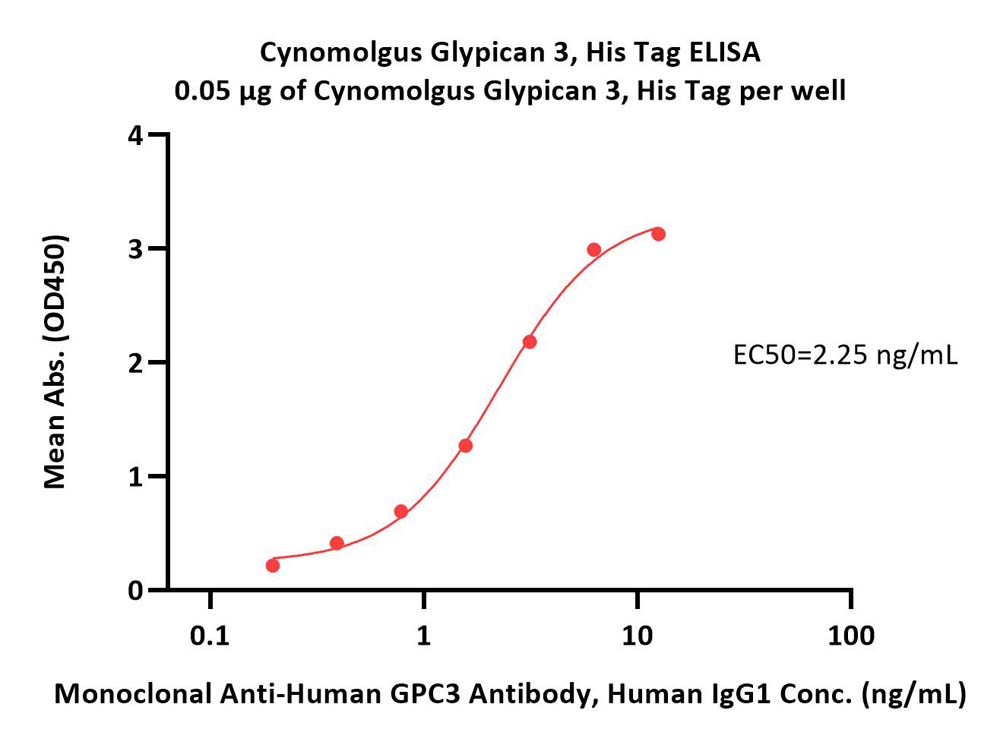
Immobilized Cynomolgus Glypican 3, His Tag (Cat. No. GP3-C5225) at 0.5 μg/mL (100 μL/well) can bind Monoclonal Anti-Human GPC3 Antibody, Human IgG1 with a linear range of 0.2-3 ng/mL (QC tested).
Protocol
背景(Background)
Glypican-3 (GPC3) is also known as Intestinal protein OCI-5, GTR2-2, MXR7, which belongs to the glypican family. Glypican 3 / GPC-3 is highly expressed in lung, liver and kidney. Glypican-3 inhibits the dipeptidyl peptidase activity of DPP4. Glypican-3 may be involved in the suppression/modulation of growth in the predominantly mesodermal tissues and organs, and also may play a role in the modulation of IGF2 interactions with its receptor and thereby modulate its function.






















































 膜杰作
膜杰作 Star Staining
Star Staining












