分子别名(Synonym)
IGHG4
表达区间及表达系统(Source)
Human IgG4 Fc, Tag Free (IG4-H5205) is expressed from human 293 cells (HEK293). It contains AA Glu 99 - Lys 327 (Accession # P01861).
Predicted N-terminus: Glu 99
Request for sequence
蛋白结构(Molecular Characterization)

This protein carries no "tag".
The protein has a calculated MW of 25.8 kDa. As a result of glycosylation, the protein migrates as 30-32 kDa under reducing (R) condition, and 28-30 kDa and 55-60 kDa under non-reducing (NR) condition (SDS-PAGE).
内毒素(Endotoxin)
Less than 1.0 EU per μg by the LAL method.
纯度(Purity)
>95% as determined by SDS-PAGE.
>95% as determined by SEC-MALS.
制剂(Formulation)
Lyophilized from 0.22 μm filtered solution in Tris with Glycine, Arginine and NaCl, pH7.5. Normally trehalose is added as protectant before lyophilization.
Contact us for customized product form or formulation.
重构方法(Reconstitution)
Please see Certificate of Analysis for specific instructions.
For best performance, we strongly recommend you to follow the reconstitution protocol provided in the CoA.
存储(Storage)
For long term storage, the product should be stored at lyophilized state at -20°C or lower.
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
- -20°C to -70°C for 12 months in lyophilized state;
- -70°C for 3 months under sterile conditions after reconstitution.
电泳(SDS-PAGE)

Human IgG4 Fc, Tag Free on SDS-PAGE under reducing (R) and non-reducing (NR) conditions. The gel was stained with Coomassie Blue. The purity of the protein is greater than 95%.
SEC-MALS
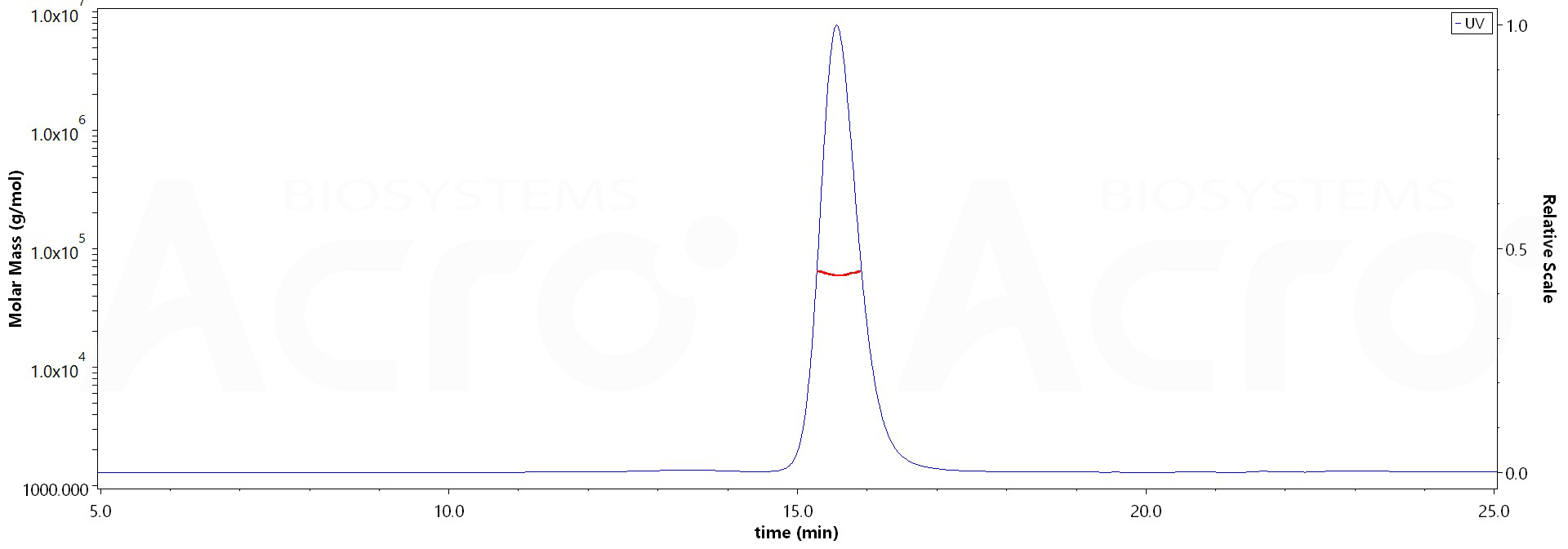
The purity of Human IgG4 Fc, Tag Free (Cat. No. IG4-H5205) is more than 95% and the molecular weight of this protein is around 54-66 kDa verified by SEC-MALS.
Report
活性(Bioactivity)-SPR
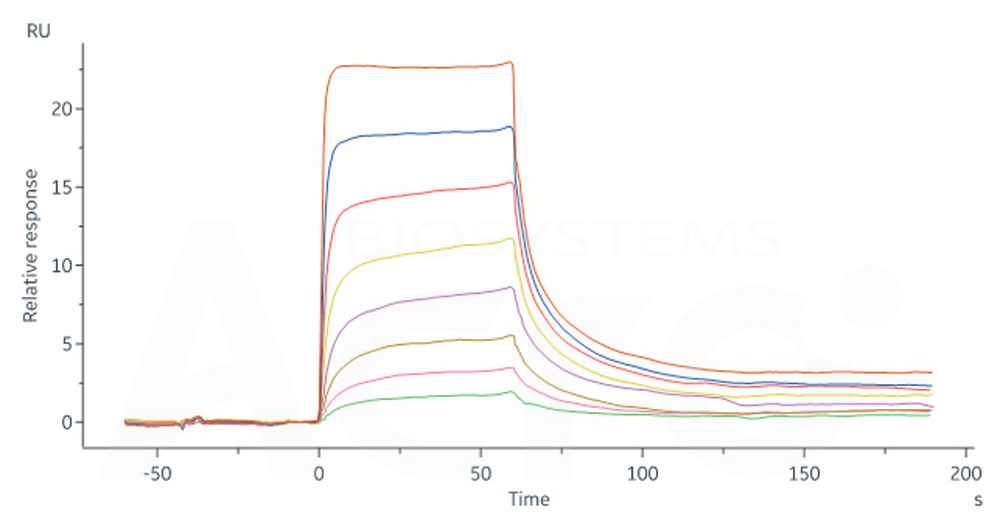
Human FCGRT&B2M Heterodimer Protein, His Tag (Cat. No. FCN-H52W7) captured on CM5 Chip via anti-His antibody can bind Human IgG4 Fc, Tag Free (Cat. No. IG4-H5205) with an affinity constant of 0.715 μM as determined in SPR assay (Biacore 8K) (QC tested).
Protocol
背景(Background)
Immunoglobulin G4 (IgG4) is a member of many immunoglobulin G developed and secreted by effective B cells. In wake of cutting by pepsin, IgG is divided into two F(ab)s with one antigen binding site and a high conserved Fc segment. The Fc segment bears a highly conserved N-glycosylation site. Ig gamma-4 chain Fc region contains two constant regions of IgG4 H chain (CH2, CH3).






















































 膜杰作
膜杰作 Star Staining
Star Staining











