分子别名(Synonym)
FGF1,AFGF,ECGF,ECGF-beta,ECGFA,ECGFB,FGF-alpha,FGFA,GLIO703,HBGF1
表达区间及表达系统(Source)
Human FGF acidic, Tag Free (AFF-H4116) is expressed from E. coli cells. It contains AA Phe 16 - Asp 155 (Accession # P05230-1).
Predicted N-terminus: Met
Request for sequence
蛋白结构(Molecular Characterization)

This protein carries no "tag"
The protein has a calculated MW of 16.0 kDa. The protein migrates as 16 kDa under reducing (R) condition (SDS-PAGE).
内毒素(Endotoxin)
Less than 1.0 EU per μg by the LAL method.
纯度(Purity)
>95% as determined by SDS-PAGE.
制剂(Formulation)
Lyophilized from 0.22 μm filtered solution in PBS, pH7.4 with trehalose as protectant.
Contact us for customized product form or formulation.
重构方法(Reconstitution)
Please see Certificate of Analysis for specific instructions.
For best performance, we strongly recommend you to follow the reconstitution protocol provided in the CoA.
存储(Storage)
For long term storage, the product should be stored at lyophilized state at -20°C or lower.
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
- -20°C to -70°C for 12 months in lyophilized state;
- -70°C for 3 months under sterile conditions after reconstitution.
电泳(SDS-PAGE)

Human FGF acidic, Tag Free on SDS-PAGE under reducing (R) condition. The gel was stained with Coomassie Blue. The purity of the protein is greater than 95%.
活性(Bioactivity)-BLI
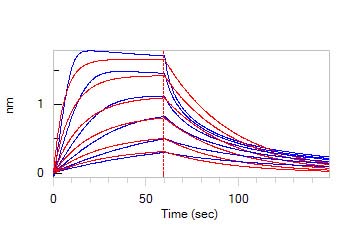
Loaded Biotinylated Heparin on SA Biosensor, can bind Human FGF acidic, Tag Free (Cat. No. AFF-H4116) with an affinity constant of 186 nM as determined in BLI assay (ForteBio Octet Red96e) (Routinely tested).
Protocol
背景(Background)
Heparin-binding growth factor 1 is a protein that in humans is encoded by the FGF1 gene. The protein encoded by this gene is a member of the fibroblast growth factor (FGF) family. FGF acidic is a potent growth factor for fibroblasts and endothelial cells. FGF acidic is involved in wound repair, angiogenesis, and development. FGF acidic is secreted from cells via an endoplasmic reticulum/Golgi independent mechanism. The ability of FGF acidic to bind to heparin sulfate is required for its ability to interact with FGF receptors and induce signaling. There are four distinct FGF receptors and each has multiple splice variants. FGF acidic binds with high affinity to many, but not all, FGFRs. Signaling cascades activated through FGF basic binding to FGFR include the ras-raf-MAPK, PLCγ/PKC, and PI3K/Akt pathways.






















































 膜杰作
膜杰作 Star Staining
Star Staining











