分子别名(Synonym)
ERBB3,HER3,LCCS2,MDA-BF-1,MGC88033,c-erbB3,erbB3-S,p180-ErbB3,p45-sErbB3,p85-sErbB3
表达区间及表达系统(Source)
Human ErbB3 Protein, Twin-Strep Tag (ER3-H5288) is expressed from human 293 cells (HEK293). It contains AA Ser 20 - Thr 643 (Accession # NP_001973.2).
Predicted N-terminus: Ser 20
Request for sequence
蛋白结构(Molecular Characterization)

This protein carries a twin strep tag at the C-terminus
The protein has a calculated MW of 71.7 kDa. The protein migrates as 85-100 kDa under reducing (R) condition (SDS-PAGE) due to glycosylation.
内毒素(Endotoxin)
Less than 1.0 EU per μg by the LAL method.
纯度(Purity)
>95% as determined by SDS-PAGE.
制剂(Formulation)
Lyophilized from 0.22 μm filtered solution in PBS, pH7.4 with trehalose as protectant.
Contact us for customized product form or formulation.
重构方法(Reconstitution)
Please see Certificate of Analysis for specific instructions.
For best performance, we strongly recommend you to follow the reconstitution protocol provided in the CoA.
存储(Storage)
For long term storage, the product should be stored at lyophilized state at -20°C or lower.
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
- -20°C to -70°C for 12 months in lyophilized state;
- -70°C for 3 months under sterile conditions after reconstitution.
电泳(SDS-PAGE)

Human ErbB3 Protein, Twin-Strep Tag on SDS-PAGE under reducing (R) condition. The gel was stained with Coomassie Blue. The purity of the protein is greater than 95%.
活性(Bioactivity)-ELISA
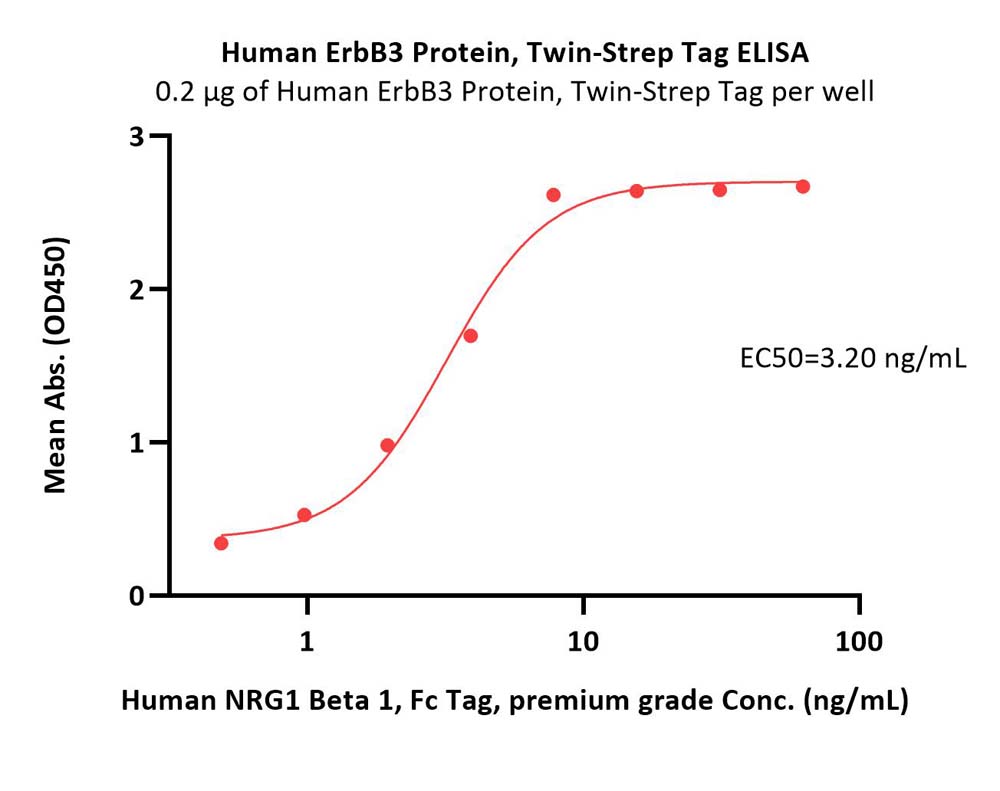
Immobilized Human ErbB3 Protein, Twin-Strep Tag (Cat. No. ER3-H5288) at 2 μg/mL (100 μL/well) can bind Human NRG1 Beta 1, Fc Tag, premium grade (Cat. No. NR1-H5268) with a linear range of 0.5-8 ng/mL (QC tested).
Protocol
背景(Background)
ErbB3,also known as Her3 (human epidermal growth factor receptor 3), is a member of the epidermal growth factor receptor (EGFR) family of receptor tyrosine kinases. This membrane-bound glycoprotein has a neuregulin binding domain but has not an active kinase domain. It therefore can bind the ligand but cannot mediate the intracellular signal transduction through protein phosphorylation. However, it does form heterodimers with ErbB2 or other EGFR members responsible for tyrosine phosphorylation to give a receptor complex and initiate the related pathway, which lead to cell proliferation or differentiation. Overexpression of this protein has been reported in numerous cancers, including prostate, bladder, and breast tumors. This protein has different isoforms derived from alternative splicing variants, and among which, the secreted isoform lacking the intermembrane region modulates the activity of membrane-bound form.






















































 膜杰作
膜杰作 Star Staining
Star Staining











