分子别名(Synonym)
CLU,Clusterin,AAG4,APOJ,CLI,KUB1,SGP-2,SP-40,TRPM2
表达区间及表达系统(Source)
Human Clusterin, His Tag (CLU-H5227) is expressed from human 293 cells (HEK293). It contains AA Asp 23 - Glu 449 (Accession # NP_001822.3).
Predicted N-terminus: Asp 23
Request for sequence
蛋白结构(Molecular Characterization)

This protein carries a polyhistidine tag at the C-terminus.
The protein has a calculated MW of 50.9 kDa. The protein migrates as 32-40 kDa under reducing (R) condition (SDS-PAGE).
内毒素(Endotoxin)
Less than 1.0 EU per μg by the LAL method.
纯度(Purity)
>95% as determined by SDS-PAGE.
制剂(Formulation)
Lyophilized from 0.22 μm filtered solution in PBS, pH7.4 with trehalose as protectant.
Contact us for customized product form or formulation.
重构方法(Reconstitution)
Please see Certificate of Analysis for specific instructions.
For best performance, we strongly recommend you to follow the reconstitution protocol provided in the CoA.
存储(Storage)
For long term storage, the product should be stored at lyophilized state at -20°C or lower.
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
- -20°C to -70°C for 12 months in lyophilized state;
- -70°C for 3 months under sterile conditions after reconstitution.
电泳(SDS-PAGE)
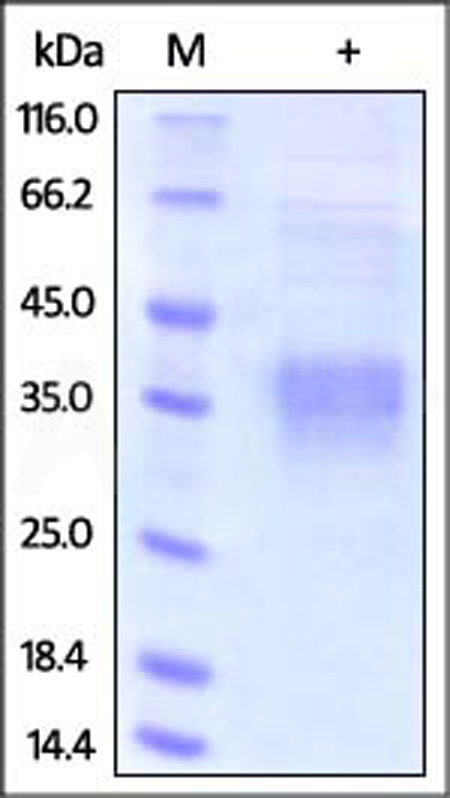
Human Clusterin, His Tag on SDS-PAGE under reducing (R) condition. The gel was stained with Coomassie Blue. The purity of the protein is greater than 95%.
活性(Bioactivity)-SPR
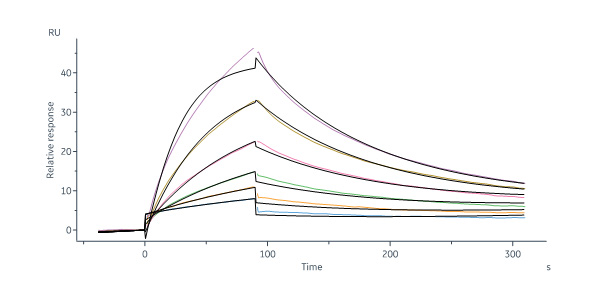
Human Clusterin, His Tag (Cat. No. CLU-H5227) immobilized on CM5 Chip can bind Human VLDL R, His Tag (Cat. No. VLR-H5227 ) with an affinity constant of 265 nM as determined in a SPR assay (Biacore 8K) (Routinely tested).
Protocol
背景(Background)
Clusterin (CLU) is also known as dimeric acidic glycoprotein (DAG protein), testosterone repressed prostate message-2 (TRPM-2), sulfated glycoprotein-2 (SGP-2) and complement lysis inhibitor (CLI), is a secreted multifunctional glycoprotein protein which belongs to the clusterin family. Intracellular cleavages of the precursor of clusterin remove the signal peptide and generate comparably sized α and β chains which are secreted as an 80 kDa Nglycosylated disulfidelinked heterodimer. DAG protein is predominantly expressed in adult testis, ovary, adrenal gland, liver, heart, and brain and in many epithelial tissues during embryonic development. Clusterin involve in several basic biological events such as cell death, tumor progression, and neurodegenerative disorders. Upregulation of clusterin mR and protein levels detected in diverse disease states and in in vitro systems have led to suggestions that it functions in membrane lipid recycling, in apoptotic cell death, and as a stress-induced secreted chaperone protein, amongst others.






















































 膜杰作
膜杰作 Star Staining
Star Staining












