产品描述(Product Details)
HRV 3C Protease is highly specific for the recognition sequence Leu-Glu-Val-Leu-Phe-Gln-↓-Gly-Pro and cleaves after the glutamine residue.
应用说明(Application)
- Removal of purification tags from fusion proteins
单位定义(Unit Definition)
One unit will cleave >95% of 100 µg control fusion protein in 50 mM Tris-HCl, 150 mM NaCl, pH 7.5 at 4℃ for 16 h.
质量控制(Quility Control)
Endotoxin : Less than 1.0 EU per μg by the LAL method.
纯度(Purity)
>95% as determined by SDS-PAGE.
制剂(Formulation)
Lyophilized from 0.22 μm filtered solution in 50 mM Tris, 150 mM NaCl, 1 mM EDTA, 1 mM DTT, pH8.0 with trehalose as protectant.
Contact us for customized product form or formulation.
存储(Storage)
This product is stable after storage at:
- -20°C to -70°C for 12 months in lyophilized state;
- -70°C for 3 months under sterile conditions after reconstitution.
电泳(SDS-PAGE)
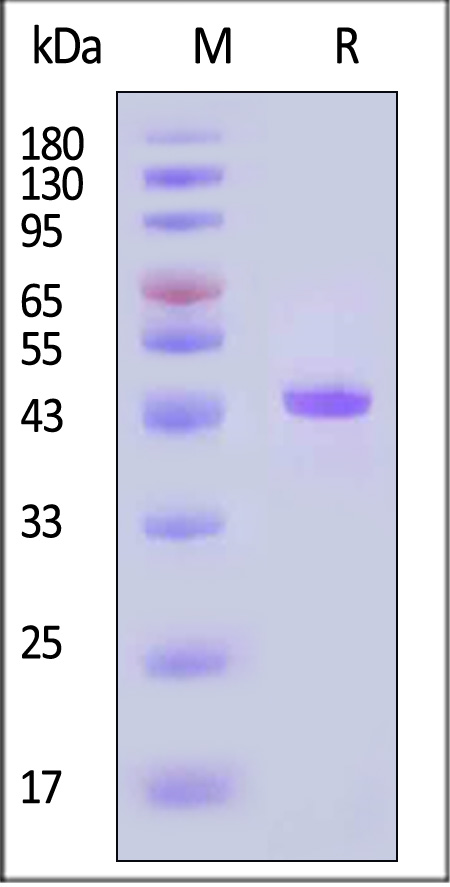
HRV-3C Protease Cleavage Enzyme, GST Tag on SDS-PAGE under reducing (R) condition. The gel was stained with Coomassie Blue. The purity of the protein is greater than 95% (With Star Ribbon Pre-stained Protein Marker).
活性(Bioactivity)
>1 Units/µg. One unit will cleave >95% of 100 µg control fusion protein in 50 mM Tris-HCl, 150 mM NaCl, pH 7.5 at 4℃ for 16 h (QC tested).






















































 膜杰作
膜杰作 Star Staining
Star Staining











