| ID | Components | Size |
| RAS008-C01 | Pre-coated SARS-CoV-2 Nucleocapsid Protein Microplate | 1 plate |
| RAS008-C02 | SARS-CoV-2 Antibody Positive Control | 100 μL |
| RAS008-C03 | SARS-CoV-2 Antibody Negative Control | 100 μL |
| RAS008-C04 | HRP-Anti-Human IgG | 200 μL |
| RAS008-C05 | 10 x Washing Buffer | 50 mL |
| RAS008-C06 | Dilution Buffer | 50 mL |
| RAS008-C07 | Substrate Solution | 12 mL |
| RAS008-C08 | Stop Solution | 7 mL |
背景(Background)
Nucleocapsid (N) protein is the most abundant protein found in coronavirus. CoV N protein is a highly immunogenic phosphoprotein important for viral genome replication and modulation of cell signaling pathways. It was first identified by a research team while they were screening for ADP-ribosylated proteins during coronavirus (CoV) infection (Grunewald M. E., et al. 2017, Virology; 517: 62-68). The array of diverse functional activities accommodated in N protein makes it more than a structural protein but also an interesting target in the development of antiviral therapeutics. Because of the conservation of N protein sequence and its strong immunogenicity, N protein of coronavirus is chosen as a diagnostic tool.
应用说明(Application)
This kit is developed for serologic test for IgG titer of Anti-SARS-CoV-2 Nucleocapsid Antibody in serum/plasma in vitro.
It is for research use only.
存储 & 运输(Storage & Shipping)
1. Unopened kit should be stored at 2℃-8℃ upon receiving.
2. Find the expiration date on the outside packaging and do not use reagents past their expiration date.
3. The opened kit should be stored per components table. The shelf life is 30 days from the date of opening.
原理(Assay Principles)
This assay kit employs a standard indirect-ELISA format, providing a rapid detection of Anti-SARS-CoV-2 antibodies in serum by SARS-CoV-2 Nucleocapsid Protein. The Kit consists of Pre-coated SARS-CoV-2 Nucleocapsid Protein Microplate, an Positive Control, an Negative Control, an HRP-Anti-Human IgG secondary antibody and related buffer.
Your experiment will include 4 simple steps:
a) Add your sample to the plate. The samples and Control sample are diluted by Dilution Buffer.
b) Add diluted Secondary antibody HRP-Anti-Human IgG to the plate. The Secondary antibody is diluted by Dilution Buffer.
c) Wash the plate and add TMB or other colorimetric HRP substrate.
d) Stop the substrate reaction by add diluted acid. Absorbance (OD) is calculated as the absorbance at 450 nm minus the absorbance at 630 nm to remove background prior to statistical analysis. The OD Value reflects the amount of antibody bound.
典型数据-Typical Data Please refer to Ds document for the assay protocol.
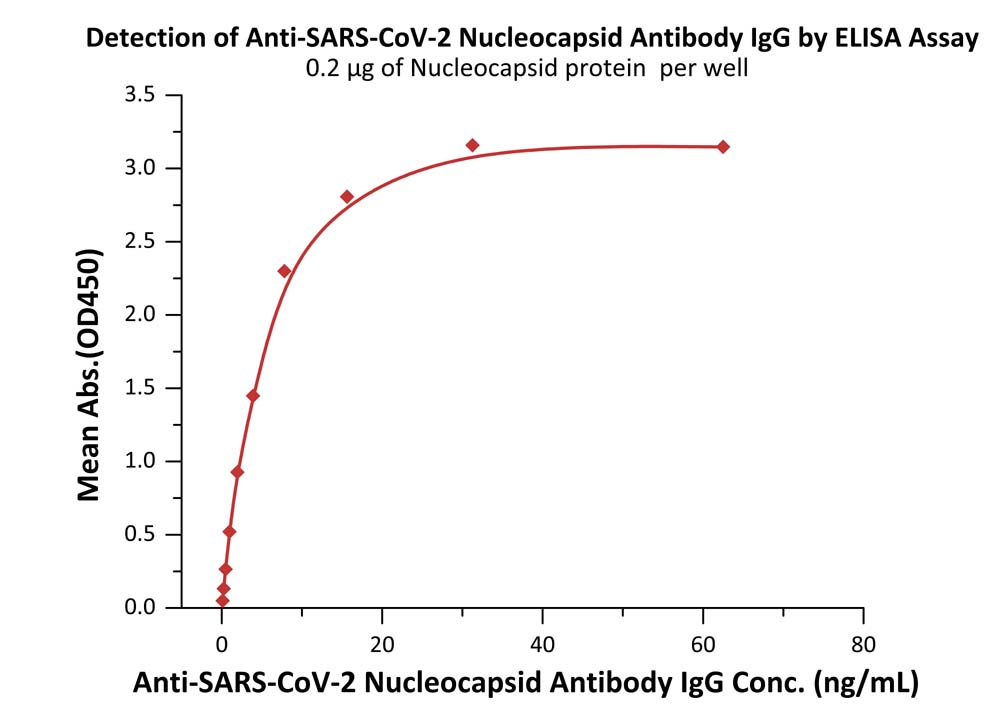
Detection of Monoclonal Anti-SARS-CoV-2 Nucleocapsid Antibody, Human IgG titer by Indirect-ELISA Assay.
Immobilized SARS-CoV-2 Nucleocapsid Protein at 2 μg/mL (100 μL/well) can bind Monoclonal Anti-SARS-CoV-2 Nucleocapsid Antibody, Human IgG in 1:400 human serum. Detection was performed using HRP-Anti-human IgG antibody with sensitivity of 0.24 ng/mL (QC tested).






















































 膜杰作
膜杰作 Star Staining
Star Staining











