抗体来源(Source)
Anti-Bevacizumab Antibodies (recommended for ADA assay) antibody is produced from a hybridoma resulting from fusion of SP2/0 myeloma and B-lymphocytes obtained from a mouse immunized with Bevacizumab.
亚型(Isotype)
Mouse IgG1/kappa
特异性(Specificity)
Recognizes Bevacizumab specifically, no cross reactivity with other humanized antibodies.
纯度(Purity)
>95% as determined by SDS-PAGE.
制剂(Formulation)
Lyophilized from 0.22 μm filtered solution in Tris with Glycine, Arginine and NaCl, pH7.5 with trehalose as protectant.
Contact us for customized product form or formulation.
重构方法(Reconstitution)
Please see Certificate of Analysis for specific instructions.
For best performance, we strongly recommend you to follow the reconstitution protocol provided in the CoA.
存储(Storage)
For long term storage, the product should be stored at lyophilized state at -20°C or lower.
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
- 4-8°C for 12 months in lyophilized state;
- -70°C for 12 months under sterile conditions after reconstitution.
电泳(SDS-PAGE)

Anti-Bevacizumab Antibody (AY9) on SDS-PAGE under reducing (R) condition. The gel was stained with Coomassie Blue. The purity of the protein is greater than 95%.
活性(Bioactivity)-ELISA
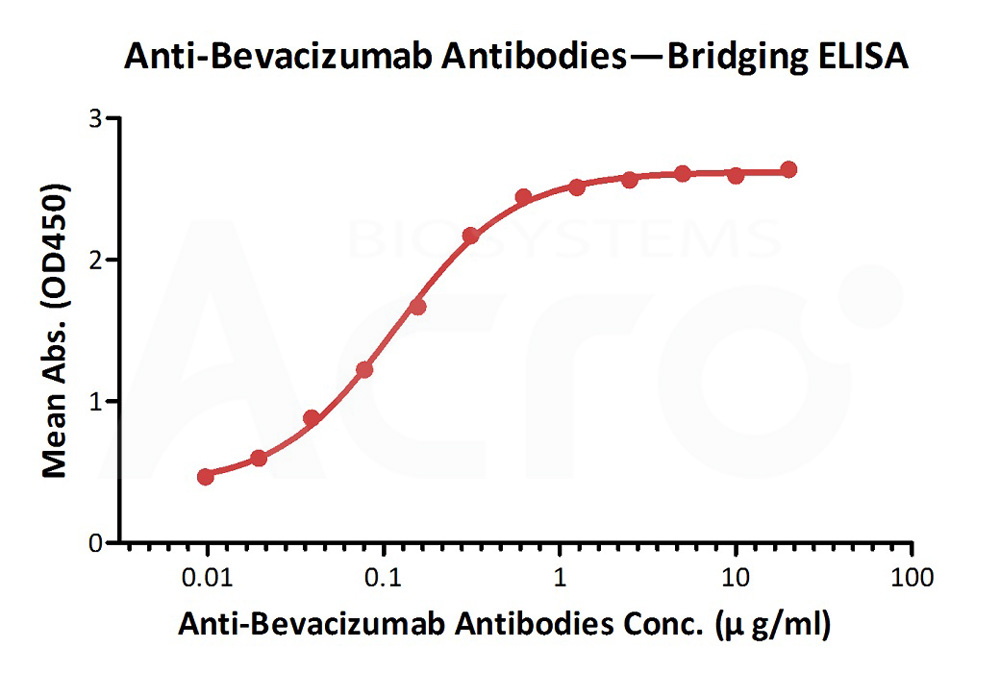
Anti-Bevacizumab Antibodies bridging ELISA for Anti-Drug Antibody (ADA) assay development. Immobilized bevacizumab at 1 µg/mL, add increasing concentrations of Anti-Bevacizumab Antibody (AY9) (Cat. No. BEB-Y9, 10% human serum) and then add biotinylated bevacizumab at 5 µg/mL. Detection was performed using HRP-conjugated streptavidin with a sensitivity of 9 ng/mL.
Protocol
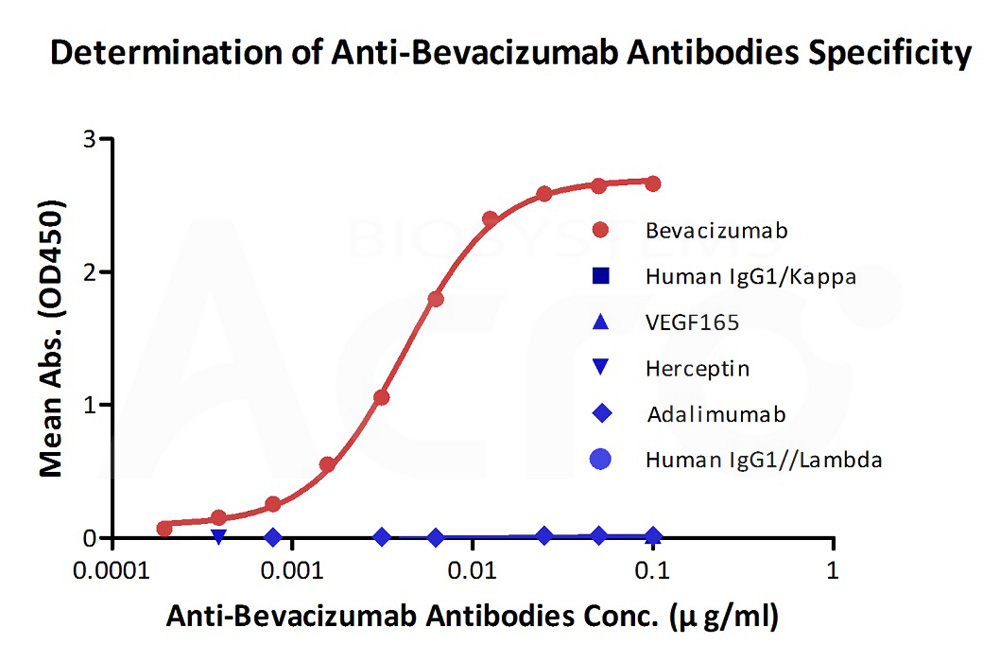
Demonstration of the specificity of Anti-Bevacizumab Antibody (AY9) (Cat. No. BEB-Y9) to the bevacizumab.
Protocol
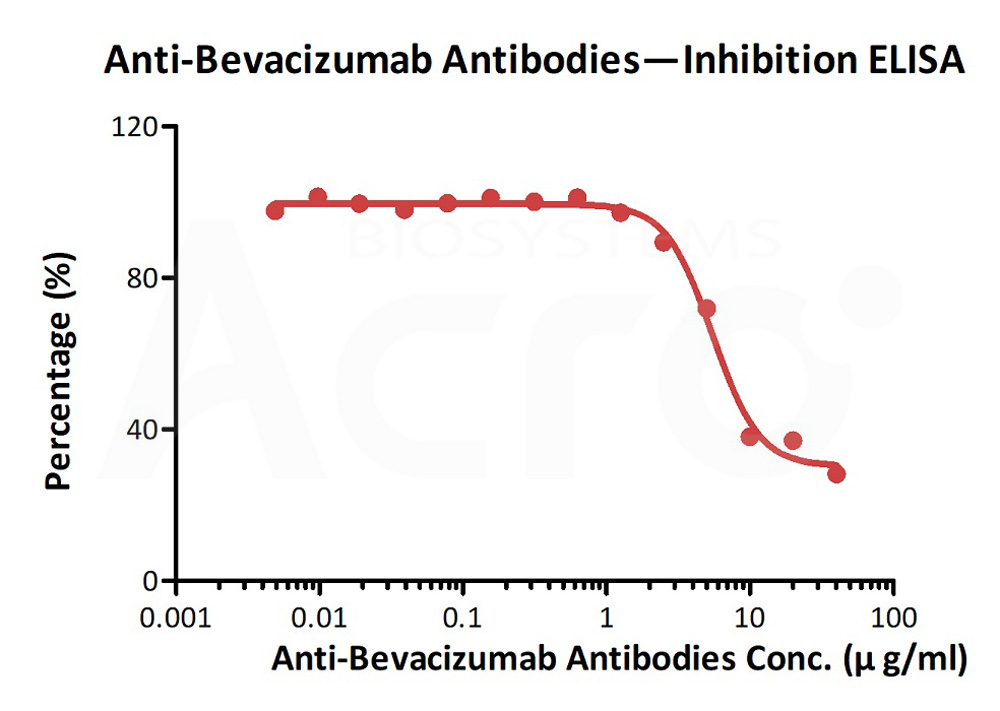
Measured by its neutrlizing ability in a functional ELISA. Immobilized bevacizumab at 2 μg/mL (100 μL/well) can bind pre-mixed Anti-Bevacizumab Antibody (AY9) (Cat. No. BEB-Y9) and Biotinylated Human VEGF165, His,Avitag (Cat. No. VE5-H82Q0) with a inhibition rate of 62%.
Protocol
活性(Bioactivity)-MSD
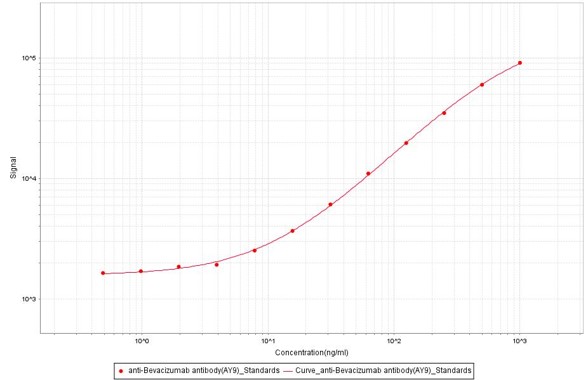
Anti-Bevacizumab Antibodies bridging MSD for Anti-Drug Antibody (ADA) assay development. Added the mix solution (biotinylated Bevacizumab at 5 µg/mL, SULFO-Bevacizumab at 5 µg/mL and increasing concentrations of Anti-Bevacizumab Antibody (AY9) (Cat. No. BEB-Y9, 100% human serum). Detection was performed using MSD Assay with a sensitivity of 0.97 ng/mL.
Protocol
活性(Bioactivity)-SPR
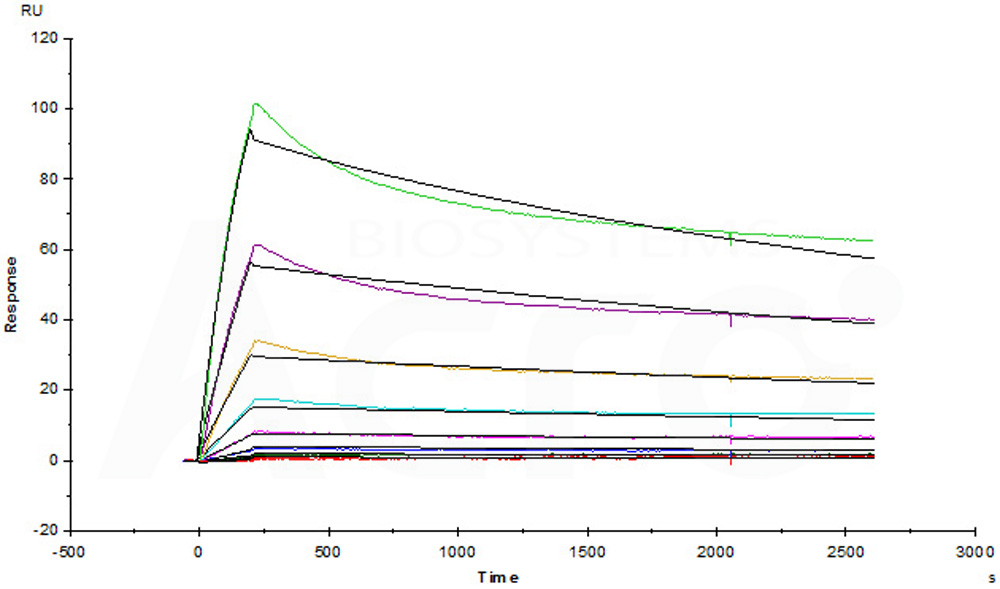
Anti-Bevacizumab Antibody (AY9) (mouse IgG1, Cat. No. BEB-Y9) captured on CM5 chip via anti-mouse antibodies surface, can bind human bevacizumab with an affinity constant of 1.92 nM.
Protocol
背景(Background)
A recombinant humanized monoclonal IgG1 antibody that binds to and inhibits the biologic activity of human vascular endothelial growth factor (VEGF). Bevacizumab contains human framework regions and the complementarity-determining regions of a murine antibody that binds to VEGF. Bevacizumab is produced in a Chinese Hamster Ovary mammalian cell expression system in a nutrient medium containing the antibiotic gentamicin and has a molecular weight of approximately 149 kilodaltons.






















































 膜杰作
膜杰作 Star Staining
Star Staining











