分子别名(Synonym)
SIGLEC10,MGC126774,PRO940,Siglec10,SLG2
表达区间及表达系统(Source)
Human Siglec-10 Protein, Mouse IgG2a Fc Tag (SI0-H525b) is expressed from human 293 cells (HEK293). It contains AA Met 17 - Thr 546 (Accession # Q96LC7-1).
Predicted N-terminus: Met 17
Request for sequence
蛋白结构(Molecular Characterization)

This protein carries a mouse IgG2a Fc tag at the C-terminus
The protein has a calculated MW of 85.6 kDa. The protein migrates as 90-120 KDa under reducing (R) condition (SDS-PAGE) due to glycosylation.
内毒素(Endotoxin)
Less than 0.1 EU per μg by the LAL method.
纯度(Purity)
>95% as determined by SDS-PAGE.
制剂(Formulation)
Lyophilized from 0.22 μm filtered solution in Tris with Glycine, Arginine and NaCl, pH7.5 with trehalose as protectant.
Contact us for customized product form or formulation.
重构方法(Reconstitution)
Please see Certificate of Analysis for specific instructions.
For best performance, we strongly recommend you to follow the reconstitution protocol provided in the CoA.
存储(Storage)
For long term storage, the product should be stored at lyophilized state at -20°C or lower.
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
- -20°C to -70°C for 12 months in lyophilized state;
- -70°C for 3 months under sterile conditions after reconstitution.
电泳(SDS-PAGE)
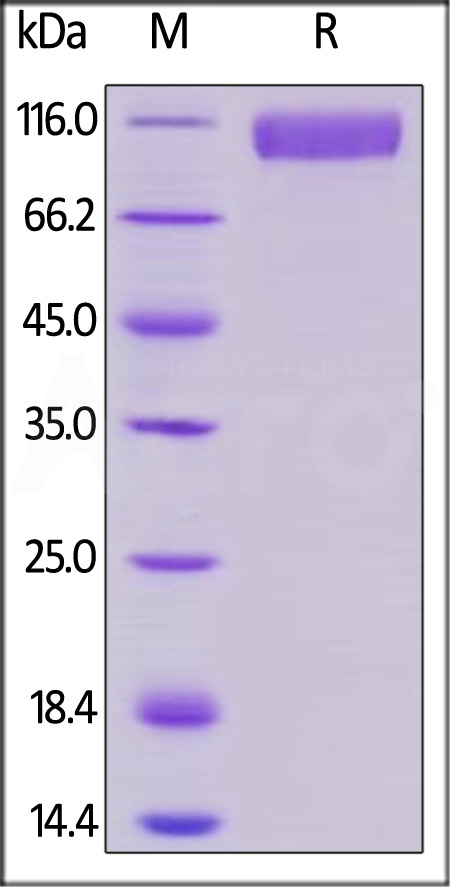
Human Siglec-10 Protein, Mouse IgG2a Fc Tag on SDS-PAGE under reducing (R) condition. The gel was stained with Coomassie Blue. The purity of the protein is greater than 95%.
活性(Bioactivity)-ELISA
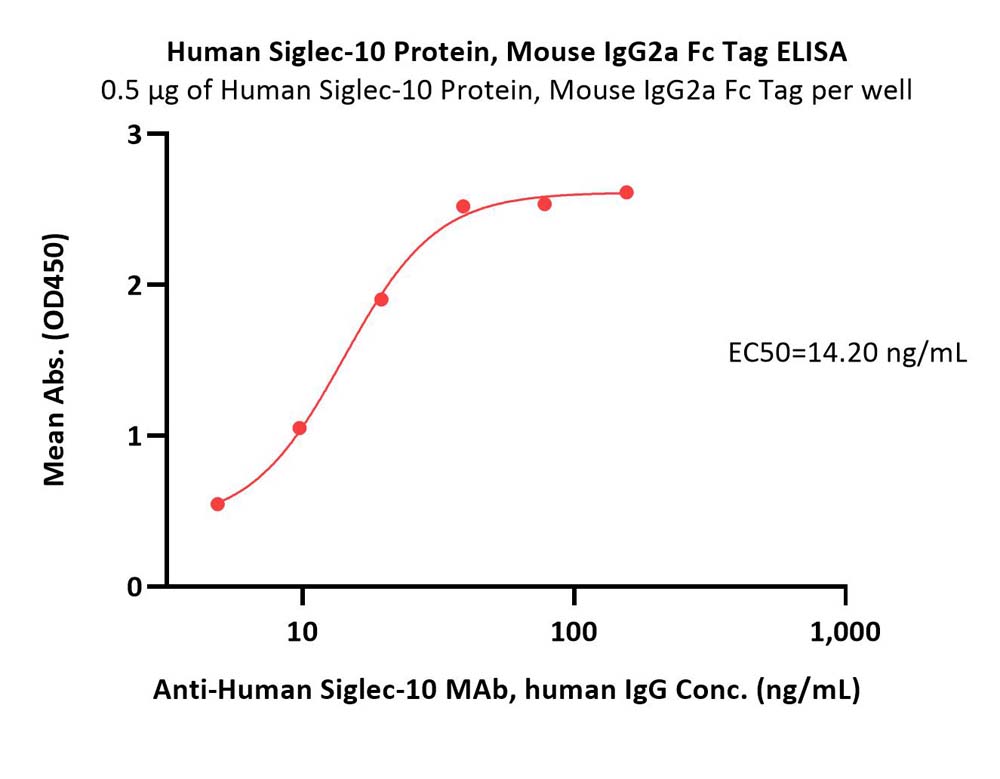
Immobilized Human Siglec-10 Protein, Mouse IgG2a Fc Tag (Cat. No. SI0-H525b) at 5 μg/mL (100 μL/well) can bind Anti-Human Siglec-10 MAb, human IgG with a linear range of 0.6-20 ng/mL (QC tested).
Protocol
背景(Background)
The siglecs (sialic acid-binding Ig-like lectins) are a distinct subset of the Ig superfamily with adhesion-molecule-like structure. We describe here a novel member of the siglec protein family that shares a similar structure including five Ig-like domains, a transmembrane domain, and a cytoplasmic tail containing two ITIM-signaling motifs. Siglec-10 was identified through database mining of an asthmatic eosinophil EST library. The Siglec-10-VAP-1 interaction seems to mediate lymphocyte adhesion to endothelium and has the potential to modify the inflammatory microenvironment via the enzymatic end products.






















































 膜杰作
膜杰作 Star Staining
Star Staining











