分子别名(Synonym)
PVR,FLJ25946,PVS,CD155,TAGE4,HVED,NECL5
表达区间及表达系统(Source)
Human CD155, Fc Tag (CD5-H5251) is expressed from human 293 cells (HEK293). It contains AA Trp 21 - Asn 343 (Accession # NP_006496.2).
Predicted N-terminus: Trp 21
Request for sequence
蛋白结构(Molecular Characterization)

This protein carries a human IgG1 Fc tag at the C-terminus.
The protein has a calculated MW of 61.7 kDa. The protein migrates as 70-90 kDa when calibrated against Star Ribbon Pre-stained Protein Marker under reducing (R) condition (SDS-PAGE) due to glycosylation.
内毒素(Endotoxin)
Less than 1.0 EU per μg by the LAL method.
纯度(Purity)
>95% as determined by SDS-PAGE.
制剂(Formulation)
Lyophilized from 0.22 μm filtered solution in Tris with Glycine, Arginine and NaCl, pH7.5 with trehalose as protectant.
Contact us for customized product form or formulation.
重构方法(Reconstitution)
Please see Certificate of Analysis for specific instructions.
For best performance, we strongly recommend you to follow the reconstitution protocol provided in the CoA.
存储(Storage)
For long term storage, the product should be stored at lyophilized state at -20°C or lower.
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
- -20°C to -70°C for 12 months in lyophilized state;
- -70°C for 3 months under sterile conditions after reconstitution.
电泳(SDS-PAGE)
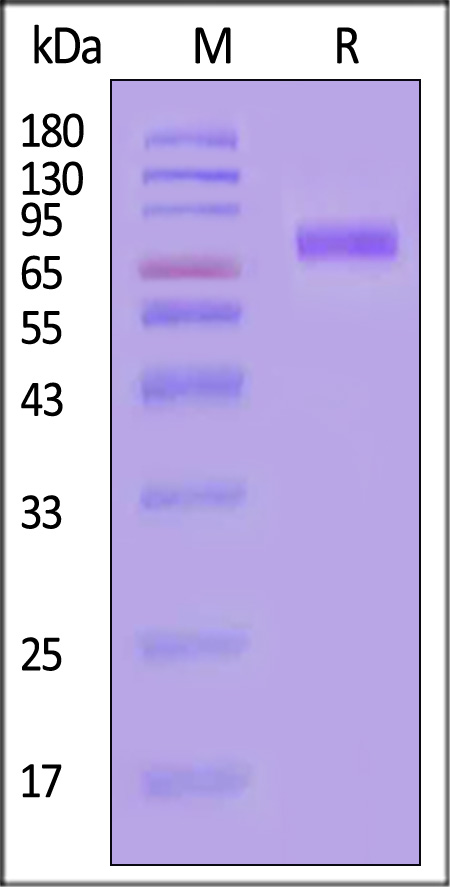
Human CD155, Fc Tag on SDS-PAGE under reducing (R) condition. The gel was stained with Coomassie Blue. The purity of the protein is greater than 95% (With Star Ribbon Pre-stained Protein Marker).
活性(Bioactivity)-ELISA
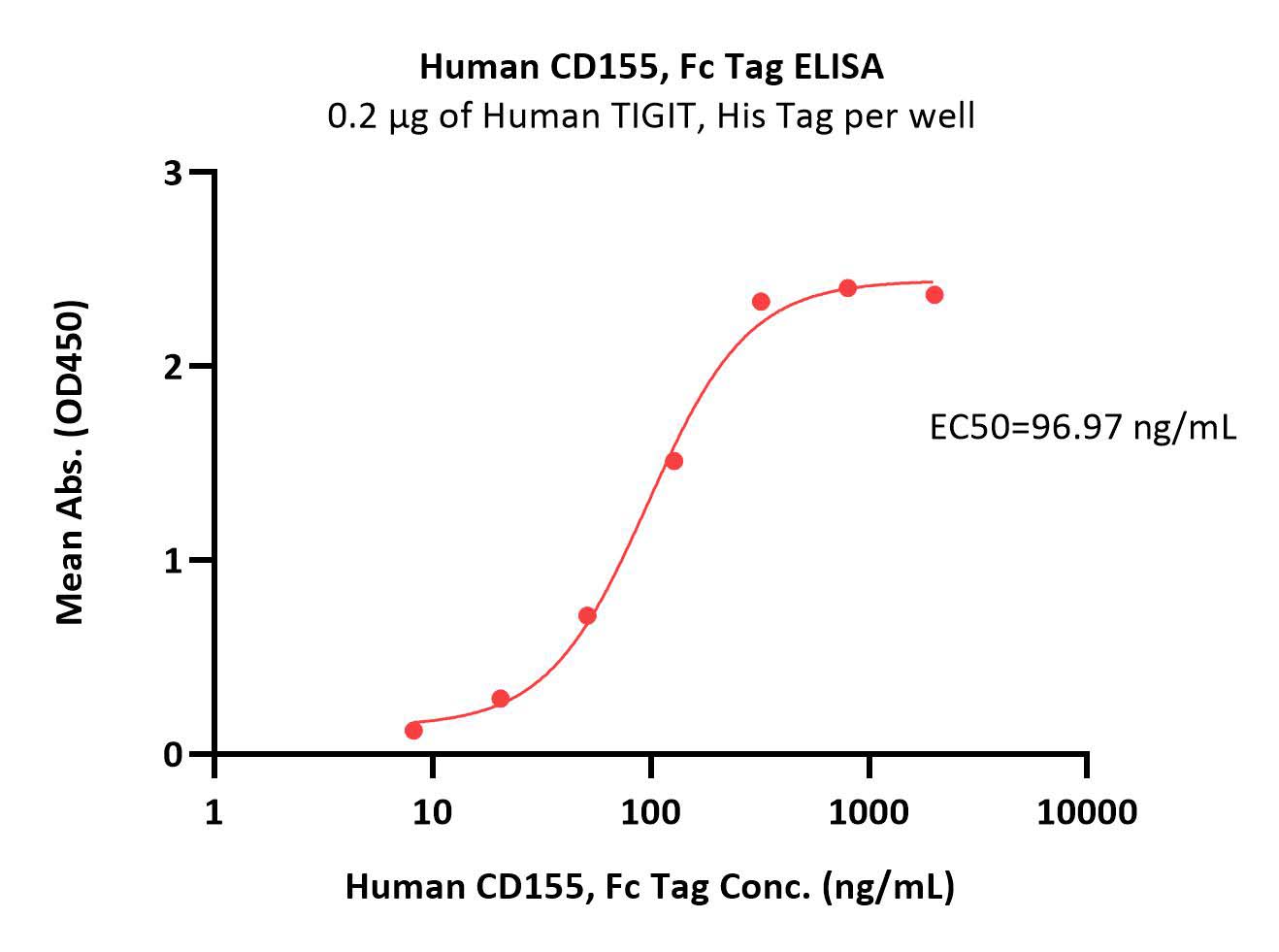
Immobilized Human TIGIT, His Tag (Cat. No. TIT-H52H3) at 2 μg/mL (100 μL/well) can bind Human CD155, Fc Tag (Cat. No. CD5-H5251) with a linear range of 8-128 ng/mL (QC tested).
Protocol
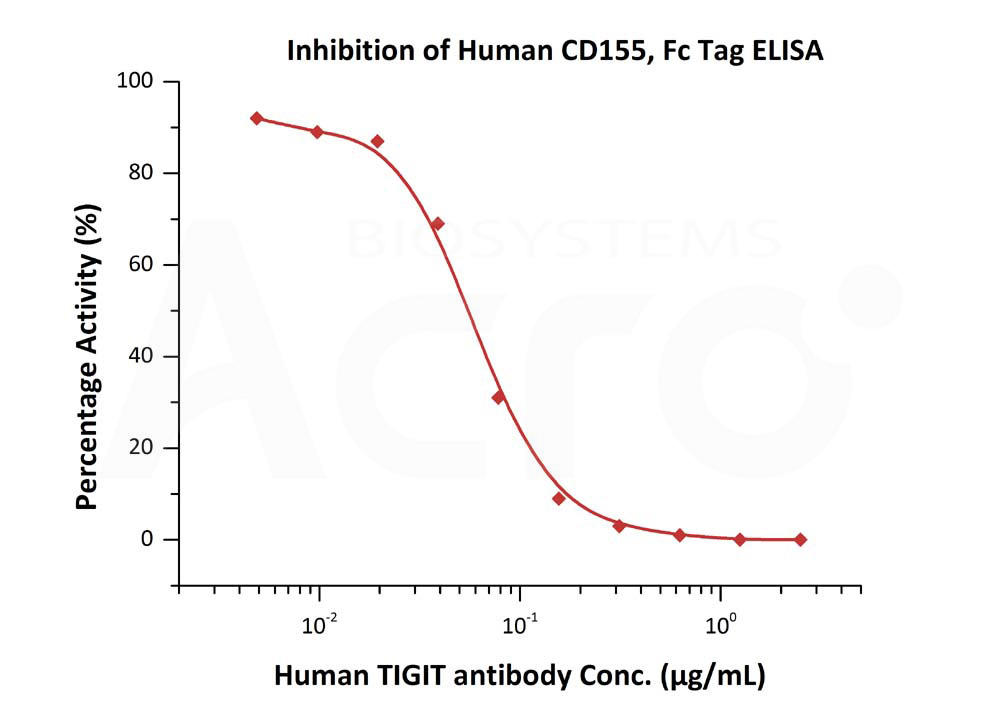
Serial dilutions of Human TIGIT Neutralizing antibody were added into Human CD155, Fc Tag (Cat. No. CD5-H5251): Biotinylated Human TIGIT, Fc,Avitag (Cat. No. TIT-H82F1) binding reactions. The half maximal inhibitory concentration (IC50) is 0.06065 μg/mL (Routinely tested).
Protocol
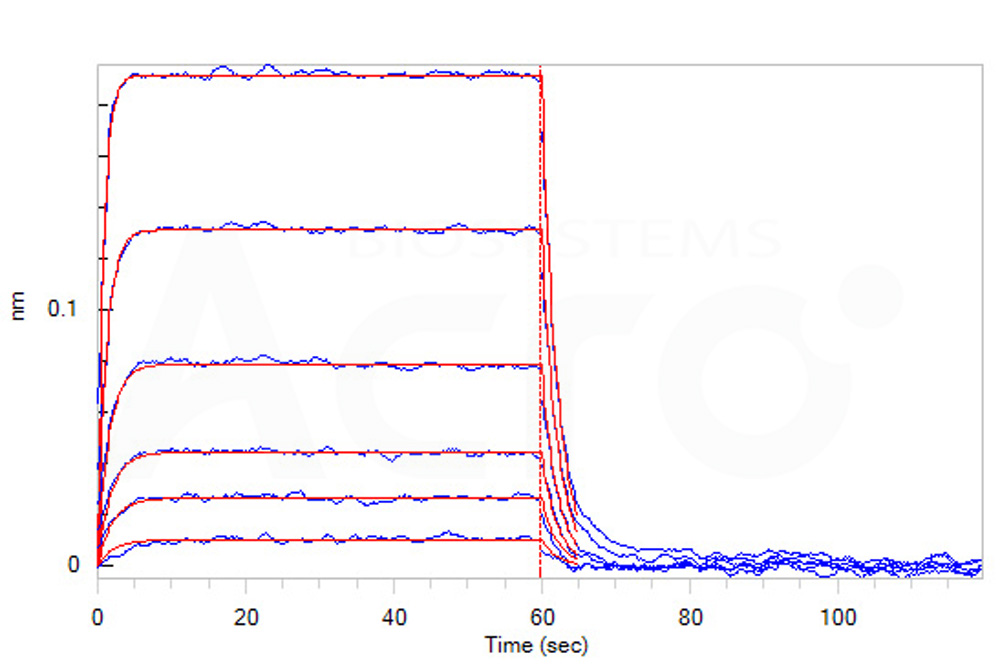
活性(Bioactivity)-BLI

Loaded Human CD155, Fc Tag (Cat. No. CD5-H5251) on Protein A Biosensor, can bind Human TIGIT, His Tag with an affinity constant of 0.23 μM as determined in BLI assay (ForteBio Octet Red96e) (Routinely tested).
Protocol
Loaded Human CD155, Fc Tag (Cat. No. CD5-H5251) on Protein A Biosensor, can bind Cynomolgus DNAM-1, His Tag (Cat. No. DN1-C52H9) with an affinity constant of 0.88 μM as determined in BLI assay (ForteBio Octet Red96e) (Routinely tested).
Protocol
活性(Bioactivity)-FACS
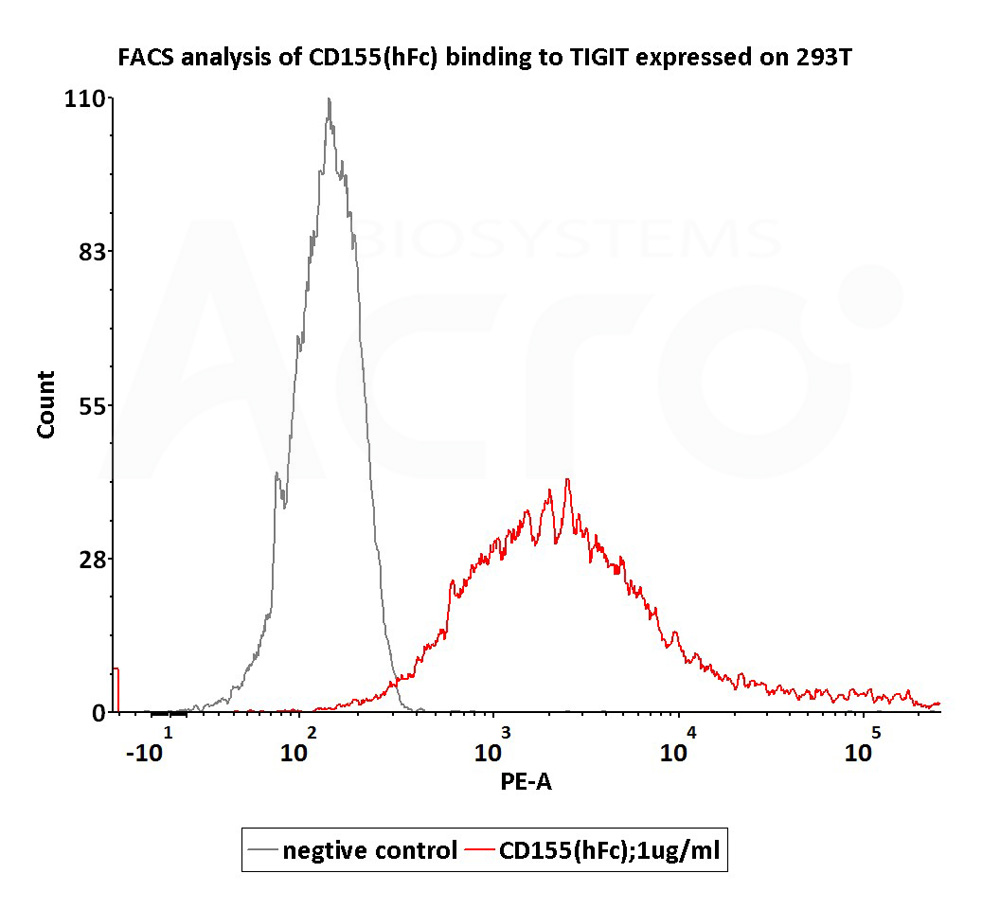
FACS assay shows that Human CD155, Fc Tag (Cat. No. CD5-H5251) can bind to 293T cell overexpressing human TIGIT. The concentration of CD155 is 1 μg/mL (Routinely tested).
Protocol
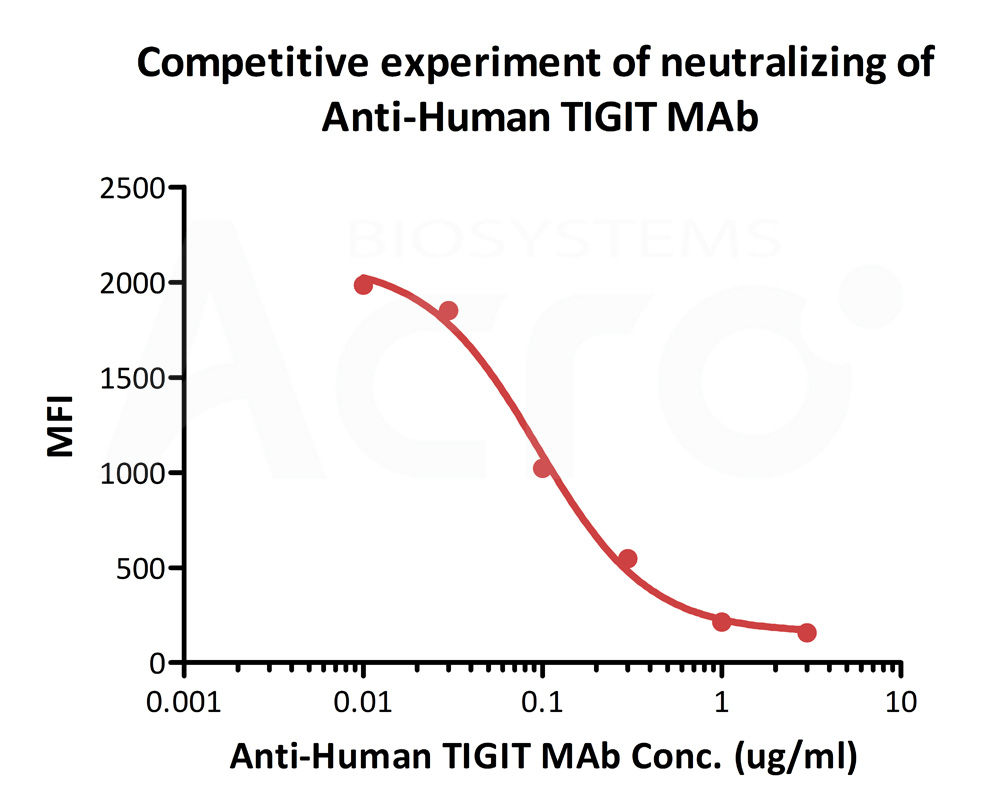
FACS analysis shows that the binding of Human CD155, Fc Tag (Cat. No. CD5-H5251) to 293T overexpressing TIGIT was inhibited by increasing concentration of neutralizing Anti-Human TIGIT MAb. The concentration of CD155 used is 1 μg/mL. The IC50 is 0.09422 μg/mL (Routinely tested).
Protocol
背景(Background)
CD155 is a Type I transmembrane glycoprotein in the immunoglobulin superfamily. Commonly known as Poliovirus Receptor (PVR) due to its involvement in the cellular poliovirus infection in primates, CD155's normal cellular function is in the establishment of intercellular adherens junctions between epithelial cells.
CD155/PVR was originally isolated based on its ability to mediate polio virus attachment to host cells. The fulllength (or CD155 alpha isoform) is synthesized as a 417 amino acid (aa) precursor that contains a 20 aa signal sequence, a 323 aa extracellular region, a 24 aa TM segment and a 50 aa cytoplasmic tail. The extracellular region contains one N terminal V type and two C2 type Ig like domains.
CD155 is a transmembrane protein with 3 extracellular immunoglobulin-like domains, D1-D3, where D1 is recognized by the virus. Low resolution structures of CD155 complexed with poliovirus have been obtained using electron microscopy while a high resolution structures of theectodomain D1 and D2 of CD155 were solved by x-ray crystallography.






















































 膜杰作
膜杰作 Star Staining
Star Staining












