分子别名(Synonym)
CD22,SIGLEC2,BL-CAM,SIGLEC-2,Siglec2,SIGLEC2FLJ22814
表达区间及表达系统(Source)
Biotinylated Human Siglec-2, Fc,Avitag (SI2-H82F8) is expressed from human 293 cells (HEK293). It contains AA Asp 20 - Arg 687 (Accession # P20273-1).
Predicted N-terminus: Asp 20
Request for sequence
蛋白结构(Molecular Characterization)

This protein carries a human IgG1 Fc tag at the C-terminus, followed by an Avi tag (Avitag™).
The protein has a calculated MW of 103.9 kDa. The protein migrates as 115-120 kDa under reducing (R) condition (SDS-PAGE) due to glycosylation.
标记(Labeling)
Biotinylation of this product is performed using Avitag™ technology. Briefly, the single lysine residue in the Avitag is enzymatically labeled with biotin.
蛋白标记度(Protein Ratio)
Passed as determined by the HABA assay / binding ELISA.
内毒素(Endotoxin)
Less than 0.2 EU per μg by the LAL method.
纯度(Purity)
>95% as determined by SDS-PAGE.
>90% as determined by SEC-MALS.
制剂(Formulation)
Lyophilized from 0.22 μm filtered solution in Tris with Glycine, Arginine and NaCl, pH7.5 with trehalose as protectant.
Contact us for customized product form or formulation.
重构方法(Reconstitution)
Please see Certificate of Analysis for specific instructions.
For best performance, we strongly recommend you to follow the reconstitution protocol provided in the CoA.
存储(Storage)
For long term storage, the product should be stored at lyophilized state at -20°C or lower.
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
- -20°C to -70°C for 12 months in lyophilized state;
- -70°C for 3 months under sterile conditions after reconstitution.
电泳(SDS-PAGE)
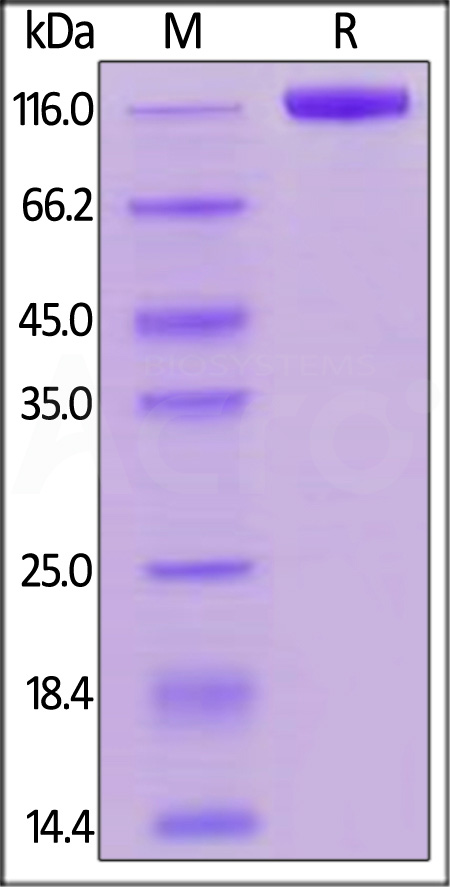
Biotinylated Human Siglec-2, Fc,Avitag on SDS-PAGE under reducing (R) condition. The gel was stained with Coomassie Blue. The purity of the protein is greater than 95%.
SEC-MALS
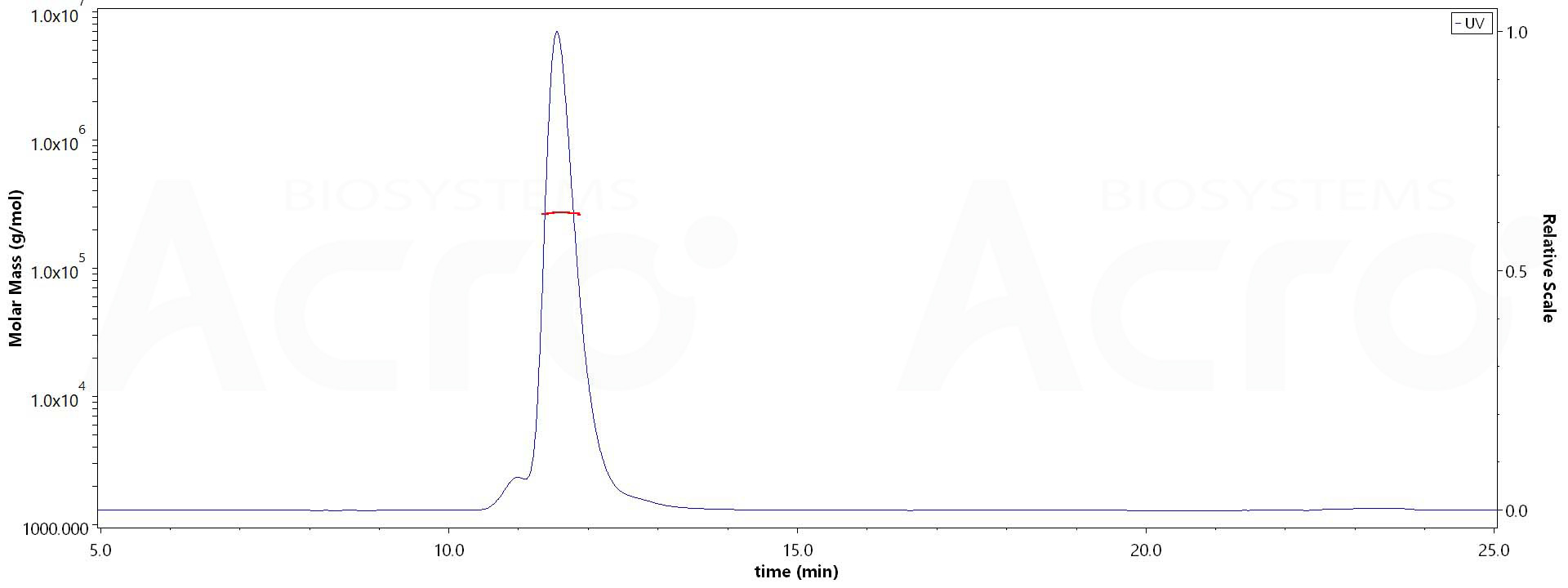
The purity of Biotinylated Human Siglec-2, Fc,Avitag (Cat. No. SI2-H82F8) is more than 90% and the molecular weight of this protein is around 250-280 kDa verified by SEC-MALS.
Report
活性(Bioactivity)-ELISA
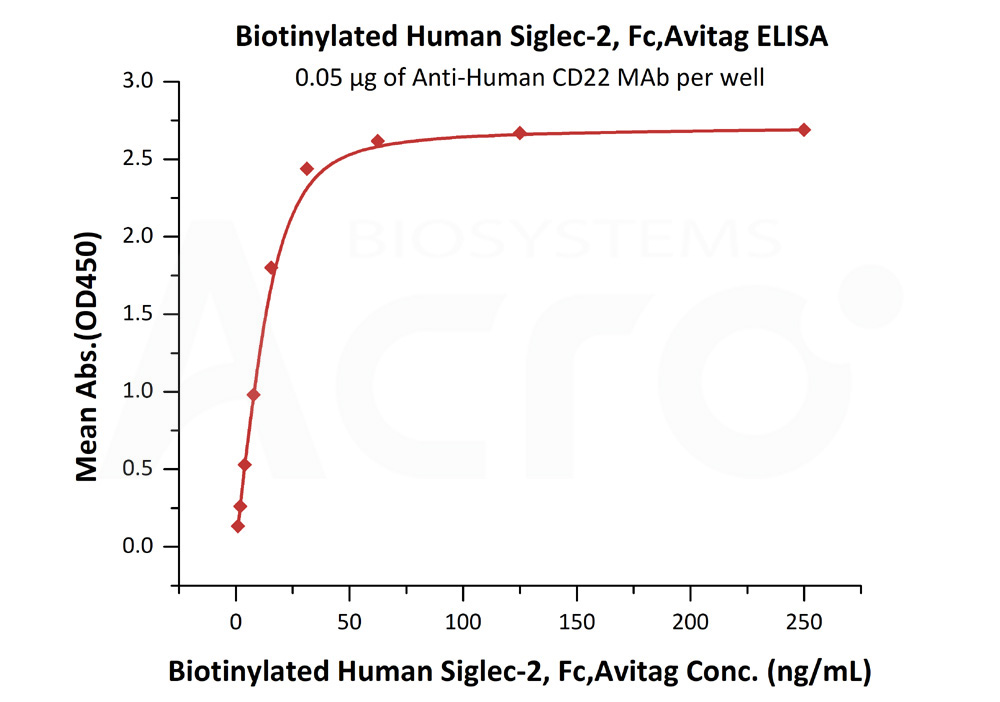
Immobilized Anti-Human CD22 MAb at 0.5 μg/mL (100 μL/well) can bind Biotinylated Human Siglec-2, Fc,Avitag (Cat. No. SI2-H82F8) with a linear range of 1-16 ng/mL (QC tested).
Protocol
CAR阳性表达率检测(Evaluation of CAR expression)
FACS Analysis of Anti-CD22 CAR Expression
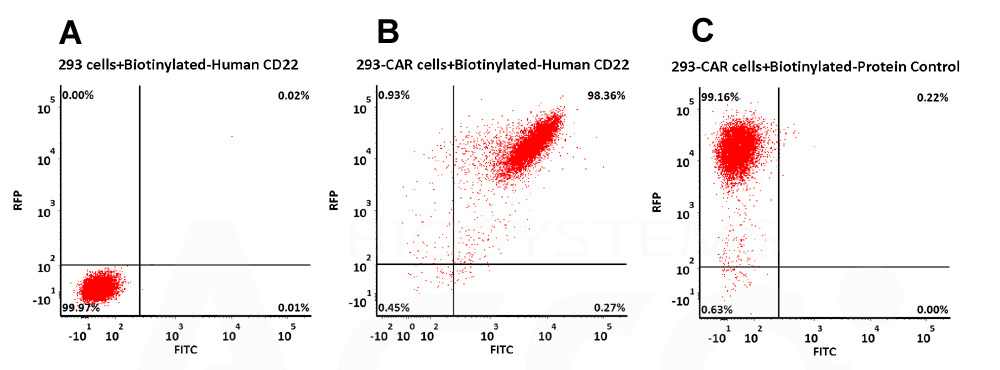
293 cells were transfected with anti-CD22-scFv and RFP tag. 2e5 of the cells were first stained with B. Biotinylated Human Siglec-2, Fc,Avitag (Cat. No. SI2-H82F8, 10 μg/mL) and C. Biotinylated Protein Control, followed by FITC Streptavidin. A. Non-transfected 293 cells and C. Biotinylated Protein Control were used as negative control. RFP was used to evaluate CAR (anti-CD22-scFv) expression and FITC was used to evaluate the binding activity of Biotinylated Human Siglec-2, Fc,Avitag (Cat. No. SI2-H82F8).
FACS Analysis of Anti-CD22 CAR Expression
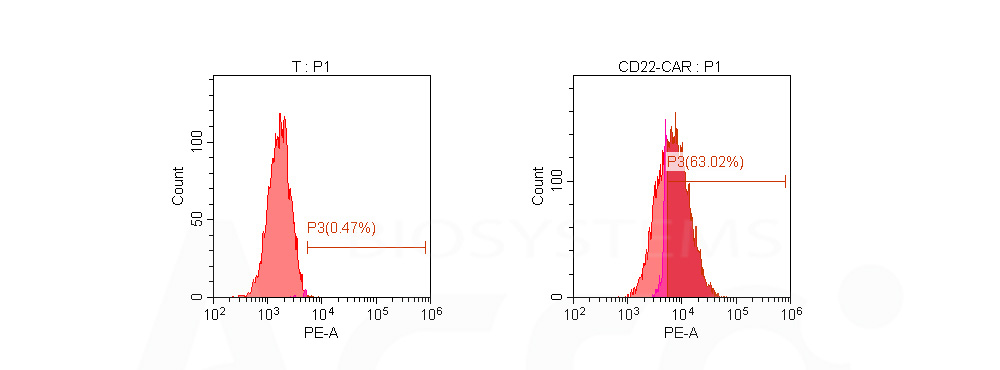
Human T cells were lentivirally transduced with anti-CD22 CAR and cultured for 3 days. Three days post-transduction, 1e6 cells were first incubated with 100 μL Biotinylated Human Siglec-2, Fc,Avitag (Cat. No. SI2-H82F8, 10 μg/mL), washed and then stained with PE Streptavidin. Non-transduced T cells were used as a control for gating of CAR expression. (Data are kindly provided by Cellyan Therapeutics Co. Ltd.)
背景(Background)
B-cell receptor CD22 is also known as Sialic acid-binding Ig-like lectin 2 (Siglec-2), B-lymphocyte cell adhesion molecule (BL-CAM), T-cell surface antigen Leu-14, which belongs to the immunoglobulin superfamily and SIGLEC (sialic acid binding Ig-like lectin) family. CD22 mediates B-cell B-cell interactions, and may be involved in the localization of B-cells in lymphoid tissues. Siglec-2 / CD22 binds sialylated glycoproteins, one of which is CD45. Siglec2 / CD22 plays a role in positive regulation through interaction with Src family tyrosine kinases and may also act as an inhibitory receptor by recruiting cytoplasmic phosphatases via their SH2 domains that block signal transduction through dephosphorylation of signaling molecules.






















































 膜杰作
膜杰作 Star Staining
Star Staining











